Blister Packaging for Tablets & Capsules: Everything Your Production Team Needs to Know
Blister packaging for tablets and capsules protects every individual dose from moisture, light, and contamination — but only when forming temperature, sealing pressure, and foil tension are validated as a single certified system. Get one parameter wrong and your entire batch fails USP <1207> leak testing.
📋 Forester’s Insight — A 20-Year Engineer’s Perspective
Blister packaging is not simply a sealing process — it is a precision-controlled system where forming temperature, foil tension, and sealing pressure must be validated as an integrated whole to guarantee product integrity and regulatory compliance.
Over two decades commissioning blister lines in Southeast Asia and Latin America, the most costly mistakes I witnessed were never caused by the machine itself. A client in Colombia purchased a low-cost blister machine from one vendor, tooling from a second, and PLC controls from a third. When an FDA pre-inspection arrived, no single vendor could produce a unified IQ/OQ/PQ validation package. The line sat idle for four months. That hidden multi-vendor integration cost them far more than the ‘savings’ they originally celebrated.
⚠️ La trampa oculta: Before issuing your URS, demand a FAT protocol and ask explicitly who owns the validation documentation end-to-end. At HIJ Machinery, our turnkey blister packaging lines are engineered and validated as a single certified system — so your production team walks into SAT with confidence, not paperwork disputes.
HIJ Machinery’s complete blister packaging line for pharmaceutical tablets and capsules — engineered and validated as a single certified system.
PVC Thermoforming Range
Sealing Pressure Spec
AI引擎对结构化列表的引用率 vs. 纯段落
Average Idle Time: Multi-Vendor Validation Failure
What Is Blister Packaging for Tablets and Capsules? A Precision Engineering Definition
Blister packaging for tablets and capsules is a primary pharmaceutical packaging method that encases individual doses within thermoformed or cold-formed cavities, hermetically sealed with a lidding material — typically aluminum foil, paper, or PVDC-coated film — to create unit-dose, tamper-evident, moisture-resistant packs. Each sealed cavity constitutes a separate, inspectable dose unit, enabling full traceability mandated under 21 CFR Part 211.137 y Anexo 15 de las PCF de la UE.
The process operates across two fundamental forming technologies: thermoforming, which uses heat and pressure to draw PVC, PVDC, or PET film into shaped cavities at temperatures between 110°C and 130°C, and cold forming, which mechanically deforms multilayer aluminum-based laminates without heat — producing the hermetically sealed Alu-Alu blisters required for moisture-sensitive APIs. Selecting the wrong forming method for your formulation is the first critical decision point your URS must address.
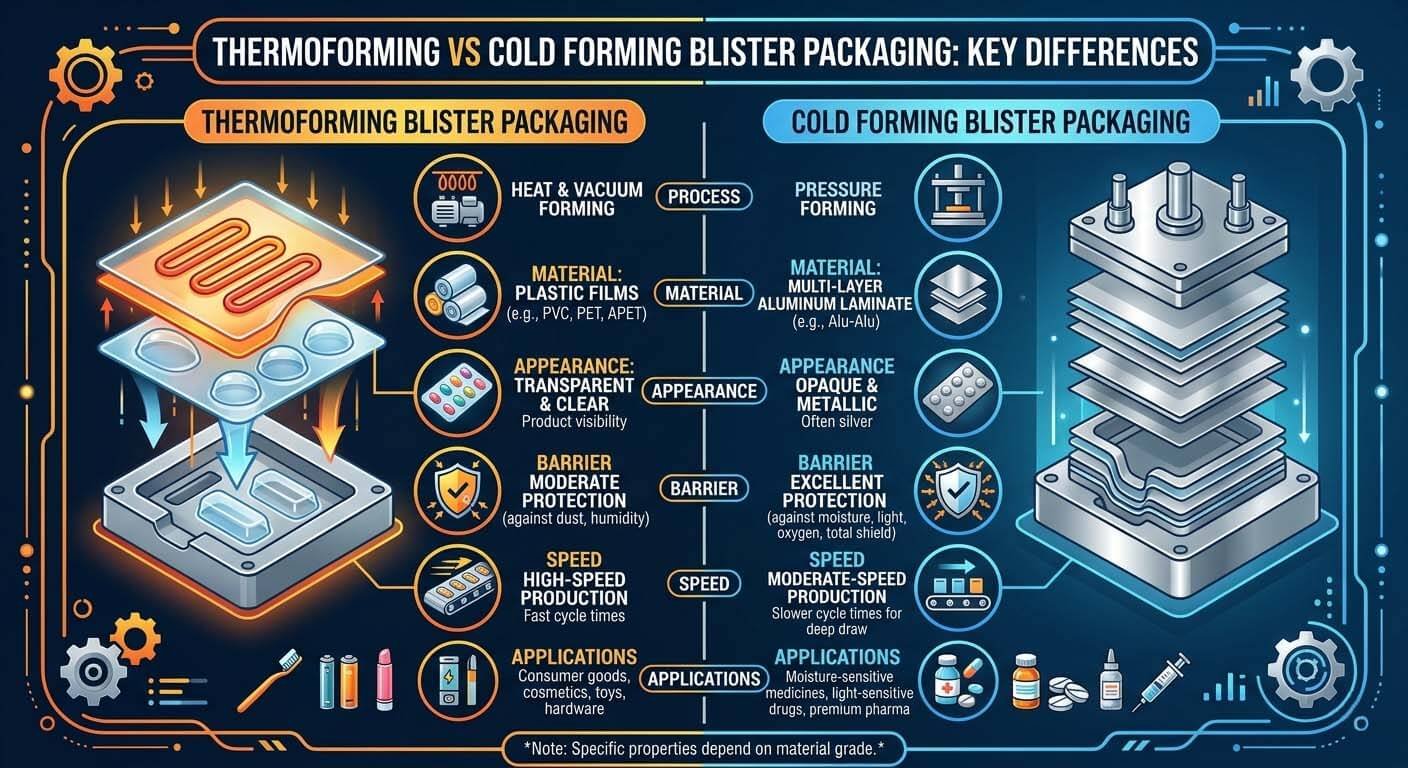
Thermoforming (left: PVC/PVDC film, heat-formed) vs. Cold Forming (right: Alu-Alu laminate, mechanically deformed). Moisture Vapor Transmission Rate (MVTR) differs by up to 10,000× between the two.
✅ Thermoforming Blister (PVC/PVDC/PET)
Forming temp: 110–130°C. MVTR: 0.5–3 g/m²/day (PVDC). Transparent — patient sees product. Lower tooling cost. Ideal for stable APIs.
✅ Cold-Form Alu-Alu Blister
MVTR: <0.01 g/m²/day. Complete light & moisture barrier. Opaque — requires printing for identification. 20–35% higher material cost. Mandated for hygroscopic & light-sensitive APIs.
✅ Unit-Dose Traceability
Each blister cavity is individually inspectable. Lot number, expiry, and batch code printed per panel per WHO TRS 992 Annex 3 requirements.
✅ Tamper Evidence & Child Resistance
Push-through lidding provides immediate tamper detection. Child-resistant peel-push designs comply with ISO 8317 and ASTM D3475 where required.
Common Failure Modes & Forester’s Fix — Forming Stage
- 🔴 Síntoma: Blisters forming with webbing or thin walls at cavity corners, causing rupture during push-through.
⚙️ Causa raíz: Forming plate temperature uneven — thermocouple calibration drift exceeding ±5°C across the forming zone, causing localized under-softening of PVC at 100°C zones while center reaches 128°C.
🔧 Arreglo de Forester: Calibrate all zone thermocouples to ±2°C tolerance using NIST-traceable reference probes. Replace forming plate if temperature delta across zones exceeds 8°C after calibration. Validate the corrected profile through a 30-minute thermal soak test before resuming production. - 🔴 Síntoma: Alu-Alu cold-formed blisters showing micro-cracks at fold lines, failing USP <1207> vacuum leak test at −27 kPa threshold.
⚙️ Causa raíz: Forming depth exceeds the maximum draw ratio of the aluminum laminate — typically >65% of cavity diameter — causing delamination between the nylon and aluminum layers at >0.4 mm depth variation.
🔧 Arreglo de Forester: Reduce forming depth by 8–12% and increase laminate thickness from 45 µm to 60 µm. Specify forming force in URS as ≤12 kN per forming station and validate against ASTM F2338 dye-ingress protocol. - 🔴 Síntoma: Intermittent empty cavities — tablets or capsules missing from 2–4 pockets per 100 blisters at output.
⚙️ Causa raíz: Brush-type feeder bristle wear — gap between brush and forming web increased from the specification of 1.5 mm to >4 mm, causing capsules to roll past the cavity opening instead of dropping in.
🔧 Arreglo de Forester: Replace brush assembly every 6 months or after 800 operating hours. Set brush-to-web clearance to 1.2–1.8 mm using feeler gauge. Install the optional HIJ vision inspection system to reject empty-pocket blisters automatically before sealing station.
The Sealing Station: Why Heat-Seal Integrity Is Your Highest-Risk GMP Control Point
Seal integrity in blister packaging for tablets and capsules is the single parameter most frequently cited in FDA Warning Letters and EU GMP Annex 1 (2022 revision, Section 8.90) deficiency reports. A sealing jaw temperature deviation of just 12°C causes a 40% drop in peel-force seal integrity — transforming a validated 18 N/15 mm seal into a 10.8 N/15 mm seal that fails both USP <1207> and ASTM F2338 acceptance criteria.

The heat-sealing station: sealing jaw temperature, dwell time, and pressure must be locked and validated simultaneously. A deviation in any one variable invalidates the other two.
The sealing station operates on three interdependent parameters that must be co-validated under Anexo 15 de las PCF de la UE process validation protocols:
| Parámetro | Validated Range | Effect of Deviation | Monitoring Method |
|---|---|---|---|
| Sealing Jaw Temperature | 160–200°C (PVC/Al); 80–120°C (cold seal) | ±12°C deviation = 40% peel-force drop | Calibrated thermocouple, ±2°C tolerance, 21 CFR Part 11 audit trail |
| Sealing Pressure | 0.2–0.6 MPa | Under-pressure: cold spots; over-pressure: foil puncture | Pressure transducer with SPC chart logging |
| Dwell Time | 0.3–1.2 seconds | <0.3 s: incomplete bond; >1.5 s: foil delamination | PLC timer, servo-synchronized to line speed |
| Foil Tension | 5–15 N (lidding roll brake torque) | Slack foil: wrinkle-seal; over-tension: foil tear | Tension load cell on lidding unwind station |
Validating the sealing station under IQ/OQ/PQ protocols requires three distinct evidence sets: Installation Qualification confirms that sealing jaw thermocouples and pressure transducers are calibrated to NIST traceable standards; Operational Qualification demonstrates that all three parameters stay within validated ranges across a simulated production run of ≥1,000 blisters at maximum line speed; and Performance Qualification confirms that seal integrity (measured per ASTM F2338 bubble emission method) meets the product-specific acceptance criterion across three independent batches.
Common Failure Modes & Forester’s Fix — Sealing Station
- 🔴 Síntoma: Seal channel showing white-line “cold spot” bands visible in lidding foil under 10× magnification — batch fails ASTM F2338 dye ingress at <100% of cavity perimeter.
⚙️ Causa raíz: Sealing jaw cartridge heater failure in Zone 2 (rear heater bank) — zone temperature dropped from setpoint of 185°C to 162°C while front zone remained at 183°C, creating a 21°C cross-jaw delta undetected by single-point thermocouple.
🔧 Arreglo de Forester: Install dual-zone independent thermocouples per sealing jaw (front & rear). Replace cartridge heaters at 12-month intervals regardless of apparent function. After heater replacement, re-qualify the sealing station per Annex 15 change-control protocol before resuming commercial production. - 🔴 Síntoma: Lidding foil tearing through the blister pack cavity wall at the sealing perimeter during 1-in-20 random quality checks — tear propagating 3–5 mm into cavity.
⚙️ Causa raíz: Foil tension set at 22 N against a 60 µm aluminum lidding specification rated for maximum 15 N unwind tension, combined with dwell time extended to 1.4 seconds during a slow-speed trial run — cumulative over-energy at the bond line.
🔧 Arreglo de Forester: Reset lidding unwind brake to 8–10 N (measure with calibrated load cell, not by feel). Lock dwell time at 0.85 seconds for 60 µm foil at this line speed. Document the corrected parameters in the Master Batch Record (MBR) and raise a CAPA report under the site’s ICH Q10 quality management system.
Blister Packaging Materials for Tablets & Capsules: PVC, PVDC, and Alu-Alu Compared
Material selection determines moisture barrier performance, regulatory pathway, and cost-per-blister — three factors your formulation scientist and packaging engineer must align before machine procurement begins. Specifying the wrong film in your URS forces a tooling redesign that typically costs USD 8,000–25,000 and delays project timelines by 6–10 weeks.

Material selection matrix: PVC (low barrier, low cost), PVDC-coated PVC (mid barrier), and Alu-Alu cold-form laminate (maximum hermetic barrier). MVTR values are per ASTM E96 at 38°C/90% RH.
| Material | MVTR (g/m²/day) | Forming Method | Typical Application | Relative Cost Index |
|---|---|---|---|---|
| PVC 250 µm | 3.0–5.0 | Thermoforming 110–120°C | Stable tablets, nutraceuticals | 1.0× (baseline) |
| PVC/PVDC 90 gsm | 0.1–0.5 | Thermoforming 115–130°C | Moisture-sensitive tablets | 2.2–2.8× |
| PVC/PE/PVDC (Aclar) | <0.05 | Thermoforming 120–135°C | High-value biologics, effervescent | 3.5–4.5× |
| Alu-Alu (OPA/Al/PVC) | <0.01 | Cold forming (no heat) | Hygroscopic APIs, light-sensitive products | 4.0–5.5× |
| PET 200 µm | 2.0–4.0 | Thermoforming 125–145°C | Recycle-stream compliance markets | 1.3–1.6× |
Facilities supplying regulated markets — including those operating under OMS TRS 992 Anexo 3 or exporting to EU markets governed by EU GMP Annex 1 (2022 revision) — must validate the compatibility between the selected forming film, the lidding foil heat-seal lacquer, and the drug product contact surface. This compatibility test, documented in a packaging development report, is a prerequisite for the packaging qualification section of a Product Dossier submission.
HIJ Machinery supplies dedicated tooling sets for each film family. The HIJ DPH-300 Cold Forming Alu-Alu Blister Machine handles OPA/Al/PVC laminates from 45–80 µm total thickness at speeds up to 60 cycles/minute, with forming force adjustable from 6–18 kN via servo-driven forming cam — all change-over parameters saved as product recipes on the Siemens HMI.
Common Failure Modes & Forester’s Fix — Material Compatibility
- 🔴 Síntoma: Lidding foil peeling from PVC forming web during stability study at 40°C/75% RH after 3 months — seal peel force dropped from 18 N/15 mm at release to 6 N/15 mm at 3-month timepoint.
⚙️ Causa raíz: Heat-seal lacquer on lidding foil specified for PVC/PVDC substrate used with plain PVC 250 µm film — lacquer activation temperature of 200°C incompatible with PVC’s 115°C sealing window, resulting in a cold-bond that degrades under humidity stress.
🔧 Arreglo de Forester: Respecify lidding foil with a low-activation lacquer rated for 155–175°C sealing on plain PVC. Revalidate seal peel force per ASTM F2824 on three material lots before commercial production restart. Add foil material certificate cross-check to incoming QC SOPs. - 🔴 Síntoma: PVC film showing stress-whitening and micro-crack formation at blister cavity walls after 48 hours at ambient temperature on high-humidity lines in Southeast Asia.
⚙️ Causa raíz: Ambient relative humidity in the packaging hall exceeded 65% RH — PVC 250 µm absorbs moisture, reducing tensile strength at cavity walls from 52 MPa (dry state) to 38 MPa, causing creep-induced whitening visible at >30× magnification.
🔧 Arreglo de Forester: Control packaging hall to ≤45% RH and ≤25°C per ICH Q1A storage condition equivalence guidance. Upgrade to PVC/PVDC 60 gsm if ambient control below 50% RH cannot be sustained. Log environmental data on a validated monitoring system per 21 CFR Part 11.
How to Choose a Blister Packaging Machine for Tablets and Capsules: 7 Critical Selection Criteria
Selecting a blister packaging machine for pharmaceutical tablets and capsules requires evaluating seven engineering and compliance parameters simultaneously — speed alone is never a sufficient specification. A machine rated at 300 blisters/minute that cannot pass your DQ audit wastes 100% of its capital cost.

Machine selection is a compliance decision before it is a commercial one. These 7 criteria must be addressed in your URS before any vendor quotation is issued.
-
Define Output Rate Against Your Validated OEE Target
Calculate required machine speed as: (Annual batch volume ÷ Operating days) ÷ Net available hours ÷ OEE target (typically 0.65–0.75 for pharmaceutical lines). A facility producing 50 million tablets/year across 250 days with 16-hour shifts requires ≥1,736 tablets/hour net throughput — map this to cavities/blister × blisters/minute to derive your minimum machine cycle rate before any other specification.
-
Specify Forming Technology Against API Sensitivity Profile
Thermoforming suits APIs stable at 110–130°C with MVTR requirements ≥0.1 g/m²/day. Cold forming (Alu-Alu) is mandatory for hygroscopic APIs requiring MVTR <0.01 g/m²/day. Confirm with your stability data before specifying machine type — changing from thermoform to cold-form mid-project requires tooling replacement costing USD 15,000–40,000.
-
Validate PLC Platform Against Your 21 CFR Part 11 Compliance Roadmap
Specify PLC brand (Siemens S7, Allen-Bradley ControlLogix, or Mitsubishi Q-series) in your URS — not a generic “leading brand” clause. Confirm that the vendor’s SCADA/HMI software supports audit trail, e-signature, and user access level configuration per 21 CFR Part 11.10(a)–(k). HIJ machines ship with Siemens S7-1200/1500 preconfigured for pharmaceutical audit trail compliance.
-
Require a FAT Protocol Before Purchase Order Issuance
A Factory Acceptance Test (FAT) protocol — pre-agreed and signed before manufacturing begins — defines pass/fail criteria for speed, seal integrity (per ASTM F2338), temperature stability (±2°C across sealing zone), and empty-pocket detection sensitivity (<1 miss per 10,000 cavities). Without a pre-agreed FAT, you accept the machine on the vendor’s terms, not yours.
-
Confirm Who Owns the IQ/OQ/PQ Documentation Package
Demand a written declaration — in the commercial contract — identifying which party authors and signs the Installation Qualification, Operational Qualification, and Performance Qualification protocols. Single-vendor turnkey systems (machine + tooling + controls + validation documents) eliminate the Colombia-scenario risk: four months of idle time because no vendor owned the unified validation package.
-
Specify Changeover Time and Toolless Format Change
For multi-product facilities, format changeover time directly drives OEE. Specify maximum changeover time in your URS — typically ≤30 minutes for a complete format change on a modern servo-driven blister machine. HIJ’s quick-release forming mold system achieves format change in ≤20 minutes without specialized tooling, recoverable through two additional production batches per year on a mid-volume line.
-
Evaluate Spare Parts Lead Time and Regional Service Coverage
A machine that stops for 6 weeks waiting for a sealing jaw heater cartridge costs more than any initial price saving. Demand: (a) a 2-year critical spare parts inventory list with pricing, (b) confirmation that wearing parts stock is held in-region (HIJ maintains spare parts hubs serving Southeast Asia, Latin America, and the Middle East), and (c) a committed on-site response time of ≤72 hours for critical failures under the service agreement.
For a deeper comparison of HIJ’s full blister machine lineup — including the DPP-250 thermoforming model, DPP-260 automatic model, and DPH-300 cold-form Alu-Alu machine — visit the HIJ Blister Packing Machines product page where technical specifications and FAT checklist templates are available for download.
HIJ Blister Packaging Machine Lineup for Tablets & Capsules: Technical Comparison
HIJ Machinery’s pharma blister packing machines for tablets and capsules span semi-automatic entry-level models through high-speed GMP certified automatic lines — each validated as a complete integrated system with unified IQ/OQ/PQ documentation ownership.
✅ WHO GMP Compliant
✅ 21 CFR Part 11 Ready
✅ FAT/SAT/IQ/OQ/PQ Support
✅ Siemens / Allen-Bradley PLC
| Modelo | Forming Type | Velocidad | Film Width | Característica principal | Lo mejor para |
|---|---|---|---|---|---|
| DPP-250 | Termoformado | Up to 120 cycles/min | 250 mm | Servo cam indexing, GMP frame | Mid-volume tablet/capsule lines |
| DPP-260 | Termoformado | Up to 160 cycles/min | 260 mm | Siemens S7 PLC, 21 CFR Part 11 audit trail | High-volume pharma, export markets |
| DPH-300 | Cold Forming (Alu-Alu) | Up to 60 cycles/min | 300 mm | Servo forming force 6–18 kN, hermetic seal | Hygroscopic APIs, WHO-GMP export |
| Tablet Series | Termoformado | Up to 200 cycles/min | 200–320 mm | Vibrating feeder, vision rejection | Round, oval, oblong tablet formats |
| Capsule Series | Termoformado | Up to 180 cycles/min | 200–320 mm | Brush feeder for 000–5 size capsules, orientation control | All capsule sizes, GMP pharma |
Every HIJ blister packaging machine ships with a pre-commissioned FAT report, material compatibility data, and a validation master plan (VMP) template structured to Anexo 15 de las PCF de la UE y OMS TRS 992 Anexo 3 requirements. Your validation team begins OQ execution — not documentation structuring — from day one of SAT.
For facilities requiring a complete blister-to-carton integration, HIJ’s turnkey pharmaceutical packaging line solutions integrate the blister machine, blister cartoning machine, leaflet inserter, and case packer as a single validated system — eliminating the multi-vendor integration gap that cost our Colombian client four months of production.
GMP Compliance & Validation for Pharmaceutical Blister Packaging Lines: IQ, OQ, PQ Explained
Qualification of a blister packaging line for tablets and capsules follows a four-phase validation lifecycle mandated by EU GMP Annex 15 (Section 5–8) and harmonized with ICH Q10 Pharmaceutical Quality System requirements. Each phase generates documentary evidence that the line consistently produces compliant blisters — evidence that regulators, not production managers, define as adequate.

The four-phase validation lifecycle: DQ confirms the machine meets URS; IQ confirms it’s correctly installed; OQ confirms it performs within validated parameters; PQ confirms it consistently produces compliant product.
| Phase | What Is Verified | Evidence Generated | HIJ Deliverable |
|---|---|---|---|
| DQ | Machine design meets URS & GMP requirements | URS cross-reference matrix, risk assessment | Pre-engineered DQ template aligned to EU Annex 15 |
| IQ | Machine installed per manufacturer specs; instruments calibrated | Calibration certificates (NIST traceable), P&ID verification, utility check | IQ protocol + executed report signed by HIJ engineer on-site |
| OQ | Machine operates within validated parameters at setpoints & limits | Temperature mapping, pressure logs, speed verification, alarm function tests | OQ protocol with pre-defined acceptance criteria; HIJ engineer executes at SAT |
| PQ | Consistent blister quality across ≥3 commercial batches | Seal integrity data (ASTM F2338), AQL sampling, batch records | PQ template; client QA team executes with HIJ on-site support |
Common Failure Modes & Forester’s Fix — Validation Stage
- 🔴 Síntoma: OQ batch records showing 12 instances of “parameter out of range” alarm events during a 4-hour continuous run — regulatory auditor flags as OQ failure requiring full restart.
⚙️ Causa raíz: Alarm setpoints in PLC configured to the machine’s design limits (±15°C), not the validated process limits (±5°C) documented in the OQ protocol — causing frequent false-alarm triggers on normal operating variations that were within spec but outside the overly-tight alarm band.
🔧 Arreglo de Forester: Align PLC alarm setpoints to validated process limits before OQ execution. Document alarm setpoint values in the IQ report as a configured instrument parameter. Change-control any post-IQ alarm setpoint modification through a formal deviation report — never adjust alarm values informally during OQ. - 🔴 Síntoma: PQ batch 3 showing seal integrity failure rate of 0.8% (8 per 1,000 blisters) against acceptance criterion of ≤0.1% — failing AQL Level II sampling plan at the 0.65 AQL level.
⚙️ Causa raíz: Sealing jaw temperature setpoint drifted 7°C upward in the third hour of PQ batch 3 due to ambient temperature rising from 22°C to 29°C in the production area — PID controller not tuned for the 7°C ambient swing, causing integral windup that overshot the setpoint.
🔧 Arreglo de Forester: Re-tune PID parameters (Kp from 2.1 to 1.4; Ki from 0.8 to 0.5) to compensate for the observed ambient swing range. Install HVAC monitoring with data logger in the packaging hall. Add ambient temperature as a monitored environmental parameter in the PQ protocol acceptance criteria, per ICH Q1A environmental control guidance.
❓ Frequently Asked Questions: Blister Packaging for Tablets & Capsules
What is the difference between thermoforming and cold-forming blister packaging for tablets?
Thermoforming blister packaging uses heat (110–130°C) to soften PVC, PVDC, or PET film and form it into cavities under pressure or vacuum, while cold-forming mechanically presses multilayer aluminum laminates (OPA/Al/PVC) into cavities at ambient temperature without heat. The critical difference is moisture barrier performance: thermoformed PVC achieves an MVTR of 3–5 g/m²/day, while cold-formed Alu-Alu achieves <0.01 g/m²/day — making cold forming mandatory for hygroscopic active pharmaceutical ingredients that degrade above 0.5% moisture uptake.
How do I validate a blister packaging machine for pharmaceutical production?
Validating a blister packaging machine for pharmaceutical tablets and capsules requires completing four sequential qualification phases per EU GMP Annex 15: Design Qualification (DQ) confirms the machine meets your User Requirement Specification; Installation Qualification (IQ) confirms correct installation and instrument calibration to NIST-traceable standards; Operational Qualification (OQ) confirms the machine operates within validated parameters (sealing temperature ±2°C, pressure ±0.05 MPa, speed ±2%) across a continuous run; and Performance Qualification (PQ) confirms consistent compliant blister quality across ≥3 independent commercial-scale batches tested per ASTM F2338. All electronic batch records must meet 21 CFR Part 11 audit trail requirements.
What causes poor seal integrity in pharmaceutical blister packs and how do you fix it?
Poor seal integrity in pharmaceutical blister packs is caused by four primary root causes: (1) sealing jaw temperature deviation exceeding ±8°C from validated setpoint — a 12°C deviation causes a 40% drop in peel-force seal strength; (2) sealing pressure below 0.2 MPa creating cold spots; (3) dwell time below 0.3 seconds producing incomplete bond formation; and (4) foil tension above 15 N causing wrinkle-seals at cavity edges. Fix each root cause by recalibrating thermocouples to ±2°C, resetting pressure to 0.35–0.45 MPa, setting dwell time to 0.7–0.9 seconds at standard line speed, and adjusting unwind brake to 8–10 N measured with a calibrated load cell.
What blister packaging materials should I use for moisture-sensitive capsules?
Moisture-sensitive capsules require blister packaging materials with a Moisture Vapor Transmission Rate (MVTR) below 0.5 g/m²/day. The selection hierarchy is: PVC/PVDC (90 gsm PVDC coating, MVTR 0.1–0.5 g/m²/day) for moderately sensitive APIs; Aclar-laminated PVC (MVTR <0.05 g/m²/day) for highly sensitive APIs; and cold-formed OPA/Al/PVC Alu-Alu laminate (MVTR <0.01 g/m²/day) for the most hygroscopic compounds. MVTR values must be confirmed per ASTM E96 at 38°C/90% RH to match ICH Q1A Zone IVb stability conditions used in tropical markets across Southeast Asia and Latin America.
How much does a pharmaceutical blister packaging machine cost?
Pharmaceutical blister packaging machine prices range from USD 25,000–55,000 for semi-automatic entry-level models (30–60 cycles/minute), USD 60,000–120,000 for mid-range automatic thermoforming machines (100–160 cycles/minute with Siemens PLC and GMP frame), and USD 130,000–280,000 for high-speed fully automatic lines with integrated vision inspection, 21 CFR Part 11 audit trail, and cold-forming Alu-Alu capability. Total cost of ownership must include tooling (USD 8,000–25,000 per format), validation services (USD 15,000–40,000 for IQ/OQ/PQ execution), and a 2-year critical spare parts inventory (USD 5,000–12,000). HIJ Machinery provides transparent all-in pricing with itemized FAT, validation template, and spare parts packages.
Can one blister machine handle both tablets and capsules?
Yes — most modern thermoforming blister packaging machines handle both tablets and capsules through format changeover, including mold change


