Blister Machine for Small Pharma & Contract Manufacturers: The Definitive Buyer’s Guide
Selecting the wrong blister machine for small pharma doesn’t just cost you capital — it costs you audit readiness, format changeover speed, and ultimately, the contracts that fund your growth. This field-tested guide cuts through the noise so you buy right the first time.
According to Forester Xiang, founder of HIJ Machinery with 20 years of production-floor experience from Bogotá to Bangkok: CMOs that purchase blister packagers without IQ/OQ documentation face re-qualification costs averaging 3× their original equipment investment when audited under FDA 21 CFR Part 211 or EU GMP Annex 1 (2022 revision).
👤 Forester’s Insight
20-Year Field PerspectiveThe Direct Answer: For small pharma and contract manufacturers, the right blister packaging machine isn’t the cheapest one — it’s the one that scales with your contract portfolio without triggering a costly compliance reset every time your SKU changes.
The Field Experience & Hidden Trap: In my 20 years of walking production floors from Bogotá to Bangkok, I’ve watched countless CMOs purchase low-cost standalone blisters with zero cGMP documentation, only to fail FDA or EU Annex 1 audits months later — absorbing re-qualification costs that tripled their original investment. The hidden trap nobody warns you about: budget machines rarely ship with IQ/OQ protocols or proper FAT/SAT support, leaving your team to ghost-write validation documents under regulatory pressure.
The Strategic Advice: Before signing any PO, demand a full URS review and confirm the supplier can support format changeover validation across multiple product types — not just the demo tablet. At HIJ, our turnkey integration philosophy means small and mid-scale buyers get the same cGMP-aligned engineering backbone as large manufacturers, without the enterprise price tag. Buy compliance once; scale confidently.
The ideal blister machine for small pharma and contract pharma blister packaging is a thermoforming or alu-alu blister packer rated 30–120 blisters/min, supplied with full IQ/OQ/PQ documentation, multi-format tooling sets, and a PLC-based audit trail compliant with 21 CFR Part 11. Compliance documentation is non-negotiable — equipment without it will fail FDA or EU GMP Annex 1 (2022) inspection, generating re-validation costs that routinely exceed $80,000 USD per audit cycle.

HIJ DPP-250 Thermoforming Pharma Blister Packaging Machine — ideal entry point for small pharma operations requiring cGMP compliance and multi-SKU format flexibility.
Why Blister Machine Selection Is a Compliance Decision — Not Just a CapEx Decision — for Small Pharma & CMOs
A contract pharma blister packaging line must do two things simultaneously: produce at viable unit economics و satisfy the regulatory inspection that follows every new client contract. Small pharma operators and CMOs — typically running 2–8 SKUs with output demands between 30,000 and 500,000 blisters per day — face a fundamentally different selection calculus than large-volume manufacturers running a single product 24/7.
The core operational reality: a CMO’s blister machine must complete validated format changeovers in under 30 minutes to stay profitable when switching between client products. Every tooling set (forming mold, sealing die, punching die) must be documented, version-controlled, and traceable under WHO TRS 992 Annex 3 and EU GMP Annex 15 requalification requirements. A machine that achieves 200 blisters/min but requires 4 hours of unvalidated changeover is a liability, not an asset.
Three machine configurations dominate the small pharma and CMO segment. Each carries distinct compliance implications:
Semi-Automatic Blister PackerOutput: 15–40 blisters/min. Lowest upfront cost ($8,000–$28,000 USD). Limited audit trail capability. Suitable for R&D-phase CMOs with <50,000 units/day. Typically requires supplemental validation documentation.
Thermoforming Automatic (PVC/PVDC)Output: 40–200 blisters/min. Mid-range cost ($25,000–$90,000 USD). Full PLC audit trail. The industry-standard platform for tablets, capsules, and pills. Most flexible format changeover profile.
Cold-Forming Alu-Alu BlisterOutput: 30–120 blisters/min. Higher unit cost ($55,000–$150,000+ USD). Mandatory for moisture-sensitive APIs requiring 0.01–0.1 g/m²·day MVTR barrier. Required by EU GMP Annex 1 for several controlled substance categories.
The Multi-SKU CMO Challenge: Why Format Changeover Validation Is Your #1 Specification
Validate the machine once for Product A. Now your new client requires Product B with a different tablet diameter (12mm vs. 9mm), a different blister format (4×7 vs. 3×5), and PVDC lidding instead of aluminum foil. Without a machine designed for rapid, documented changeover, you face a full requalification cycle under EU GMP Annex 15 — costing 3–6 weeks and $15,000–$40,000 per change depending on your regulatory market. HIJ’s DPP-250 and DPP-260 series are engineered with tooling-locking mechanisms that achieve format changeover documentation in under 2 hours, using numbered tooling sets that map directly to your URS deviation log.

cGMP-compliant blister packaging operations require both machine-side audit trail capability and operator documentation protocols — both must be specified in your URS.
Common Failure Modes & Forester’s Fix — Machine Selection Phase
- 🔴 Symptom: CMO purchases low-cost blister packer; fails EU GMP Annex 1 audit 8 months later due to absent sealing temperature log and missing IQ documentation.
⚙️ Root Cause: Machine’s PLC uses a proprietary, non-exportable data format with no 21 CFR Part 11-compliant audit trail. Sealing jaw temperature controller lacks calibration traceability (tolerance drift >±5°C undetected).
🔧 Forester’s Fix: Specify at minimum a Siemens S7-1200 or Allen-Bradley ControlLogix PLC with CSV-exportable batch records. Sealing jaw temperature tolerance must be ≤±2°C with 4-hour calibration cycle logging. Confirm IQ/OQ template availability before PO signature. - 🔴 Symptom: Format changeover from 10×1 tablet blister to 5×2 capsule blister takes 6+ hours; CMO loses $12,000 in production time per switch.
⚙️ Root Cause: Tooling sets are not numbered or version-controlled; forming mold alignment requires manual shimming with no repeatability reference points.
🔧 Forester’s Fix: Require machines with indexed tooling holders (dowel-pin alignment) and laser-etched tooling serial numbers that map to the validated change-part catalogue. Target changeover time: <45 minutes from last good blister to first good blister of new format, fully documented. - 🔴 Symptom: Sealing integrity failures at 15–18% rate on humid-climate production sites (Southeast Asia, Latin America), causing product recalls.
⚙️ Root Cause: Sealing station heat distribution uneven across jaw width (>8°C differential from center to edge); inadequate foil tension control causing micro-wrinkles in lidding film.
🔧 Forester’s Fix: Specify multi-zone sealing temperature control with ≤3°C cross-jaw uniformity. Foil tension servo control (0.5–5.0 N adjustable) prevents lidding wrinkle. Validate per ASTM F2338 (non-destructive package integrity testing) with 100% vacuum decay test records.
Critical Technical Specifications for Contract Pharma Blister Packaging Machines: What Your URS Must Cover
Specifying a blister packer without a User Requirements Specification (URS) is the fastest route to a failed Qualification (IQ/OQ/PQ) cycle. The URS bridges your regulatory obligations to the machine’s technical capabilities — and for CMOs, it must account for your broadest possible product portfolio, not just your current client list. The 9 technical parameters below represent the minimum viable specification for any pharma-grade blister machine intended for FDA or EU GMP regulated markets.
Thermoforming vs. Cold-Forming Alu-Alu: Choosing the Right Platform for Your API Portfolio
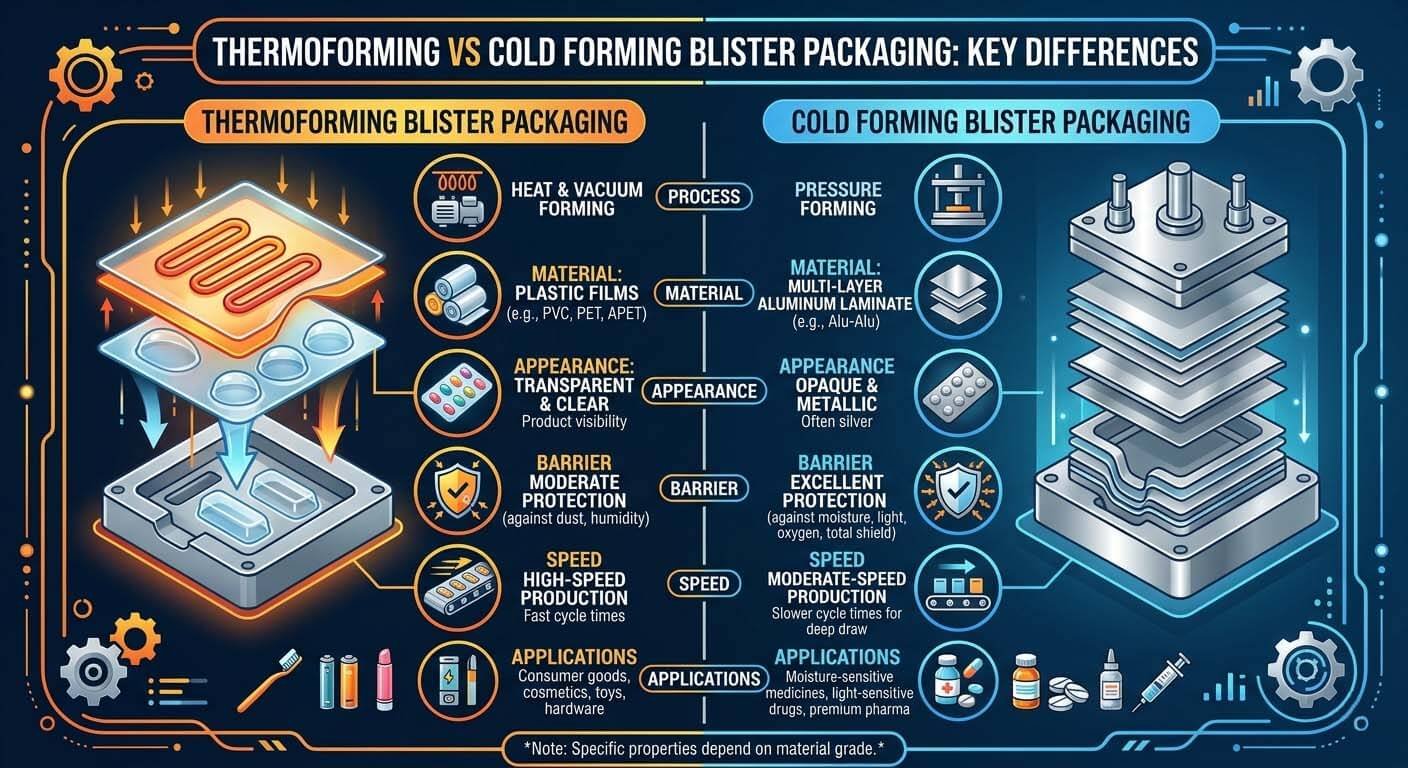
Thermoforming (PVC/PVDC) vs. Cold-Forming Alu-Alu: the barrier performance gap determines which platform your API portfolio demands.
| Parameter | HIJ Thermoforming (DPP-250/260) | HIJ Cold-Forming Alu-Alu (DPH-300) | Generic Budget Machines |
|---|---|---|---|
| Output Speed | 40–200 blisters/min | 30–120 blisters/min | 20–80 blisters/min |
| Forming Film | PVC, PVDC, PVC/PE, PP | Alu/PVC/PE (cold-form foil) | PVC only (most models) |
| Moisture Barrier (MVTR) | 0.3–3.0 g/m²·day (PVDC) | <0.01 g/m²·day (superior) | Unspecified / unlisted |
| Sealing Temp Control | Multi-zone ±2°C | Multi-zone ±2°C | Single-zone ±8–15°C |
| PLC / Audit Trail | Siemens S7 / Allen-Bradley; 21 CFR Pt 11 ready | Siemens S7 / Allen-Bradley; 21 CFR Pt 11 ready | Proprietary; no export function |
| IQ/OQ/PQ Docs | Full FAT/SAT + IQ/OQ/PQ templates | Full FAT/SAT + IQ/OQ/PQ templates | None standard; quotes extra |
| Format Changeover | <45 min with indexed tooling | <60 min with indexed tooling | 3–8 hours; no repeatability |
| CE / WHO GMP | CE certified; WHO GMP aligned | CE certified; WHO GMP aligned | CE claimed; no documentation |
| Sealing Integrity Test | ASTM F2338 / USP <1207> compatible | ASTM F2338 / USP <1207> compatible | Visual only; no instrument data |
| نطاق السعر (بالدولار الأمريكي) | $28,000–$75,000 | $55,000–$130,000 | $6,000–$22,000 |
Explore the full HIJ Blister Packing Machine product line including thermoforming, alu-alu, liquid blister, and capsule blister variants — all engineered to cGMP standards.
The 9 Non-Negotiable URS Parameters for CMO Blister Packaging Equipment
- Sealing jaw temperature uniformity ≤±2°C — A 12°C deviation in jaw temperature causes a 40% drop in seal integrity across the blister width, per ASTM F2338 validation data. Multi-zone PID control is mandatory.
- 21 CFR Part 11-compliant electronic batch records — Requires time-stamped, operator-attributed, tamper-evident data logs for every production run. CSV or SQL export mandatory for regulatory submission.
- Validated changeover time ≤60 minutes for all format combinations documented in your product catalogue — measured from last good blister (Format A) to first good blister (Format B), fully traceable.
- MVTR specification for forming film — Minimum: PVC ≤3.0 g/m²·day; PVDC ≤0.3 g/m²·day; alu-alu ≤0.01 g/m²·day. Must be supported by film supplier certificate of analysis in DHF.
- Reject system with 100% cavity inspection — Camera or weight-based empty-cavity detection with automatic rejection; rejection events logged to batch record per 21 CFR Part 211 Subpart K.
- GMP-grade stainless steel contact surfaces — SS 316L for all product-contact parts; Ra ≤0.8 μm surface finish; crevice-free welds per ASME BPE standard.
- FAT (Factory Acceptance Test) protocol — Minimum 4-hour witnessed run at specified speed with <0.5% defect rate; all alarm and safety interlock functions tested and documented.
- IQ/OQ/PQ template package — Supplier-provided master templates aligned to EU GMP Annex 15 and ICH Q10 pharmaceutical quality system requirements. Templates must be editable and site-customizable.
- Spare parts availability guarantee — minimum 10 years — Confirmed in writing, including critical wear parts: forming mold, sealing die, punching tool, sealing gasket, cam follower.
EU GMP Annex 15
21 CFR Part 211
21 CFR Part 11
WHO TRS 992 Annex 3
ICH Q10
ASTM F2338
USP <1207>
ISO 13849-1
Common Failure Modes & Forester’s Fix — Technical Specification Phase
- 🔴 Symptom: Blister seal fails vacuum decay test (USP <1207> Method B) at 8% defect rate after 3 months of operation — auditor flags as critical deviation under 21 CFR Part 211.134.
⚙️ Root Cause: Sealing gasket hardness has drifted from Shore A 55 to Shore A 72 due to heat cycling; combined with lidding foil tension inconsistency (±18% measured), causing micro-channel seal defects invisible to visual inspection.
🔧 Forester’s Fix: Implement 500-hour PM schedule for sealing gasket replacement. Specify gasket material as silicone (HTV grade, Shore A 50–60 at operating temperature). Install foil tension load cell (target: 2.5 N ± 0.3 N) with real-time batch record logging. Re-qualify per USP <1207> Method B after each gasket change. - 🔴 Symptom: Tablets fractured in 3–5% of blisters during high-speed forming at 160 blisters/min; customer complaint triggers FDA 483 observation.
⚙️ Root Cause: Forming heating plate temperature set at 180°C on a PVC film rated for 155–165°C; over-softened film collapses partially under tablet weight during cavity transfer, causing tablet-edge impact fractures.
🔧 Forester’s Fix: Recalibrate forming station to 158°C ± 2°C for 250 μm PVC film. Verify with IR thermometer across full forming width (9 measurement points). Enable tablet counting rejection for partial blisters; log all rejection events per 21 CFR Part 211. - 🔴 Symptom: PLC batch records cannot be exported or printed in auditable format — FDA inspector issues 483 observation for 21 CFR Part 11 non-compliance.
⚙️ Root Cause: Machine ships with a proprietary touchscreen HMI that stores data in binary format; no USB export or network connectivity; no role-based access control or e-signature capability.
🔧 Forester’s Fix: Specify Siemens S7-1500 or Allen-Bradley ControlLogix PLC with WinCC SCADA interface. Require: (1) role-based login with minimum 3 access levels, (2) time-stamped CSV export with digital checksum, (3) 30-day local data retention minimum, (4) supplier-provided 21 CFR Part 11 compliance matrix document before PO.
IQ/OQ/PQ Validation for CMO Blister Packaging Lines: What Your Supplier Must Deliver
Validation is the process by which your team — and your regulatory authority — confirm that a blister machine consistently produces output meeting predetermined quality specifications. For CMOs operating under contract pharma blister packaging agreements, the validation burden is amplified: each new client product typically triggers a requalification event, and a machine that wasn’t designed for documented changeover will generate validation costs that destroy your project margin within 18 months.
The DQ → IQ → OQ → PQ sequence is not bureaucratic overhead — it is your liability shield. Under EU GMP Annex 15 (2015) and 21 CFR Part 211 Subpart J, a missing or incomplete IQ (Installation Qualification) record renders every subsequent batch manufactured on that equipment potentially non-releasable. Here is what each stage must document for a blister packaging line:

The DQ → IQ → OQ → PQ validation lifecycle: every blister packaging machine entering a regulated CMO facility must complete this sequence before commercial batch release.
The DQ → IQ → OQ → PQ Validation Roadmap for Blister Packaging Equipment
-
DQ — Design Qualification: Lock Your URS Before OrderingDocument that the machine design meets your regulatory and operational requirements before purchase. DQ deliverables: completed URS, supplier DQ checklist signed by qualified person, risk assessment per ICH Q9 covering sealing integrity, product contact materials, and data integrity. Confirm alu-alu vs. thermoforming platform decision here — changing platforms post-DQ triggers full revalidation.
-
FAT — Factory Acceptance Test: Qualify at the Supplier’s FloorConduct a witnessed, minimum 4-hour production run at the supplier’s facility at your specified output speed. FAT punch list must include: all alarm testing (low film, misaligned blister, temperature deviation >5°C), sealing integrity sample pull (minimum 30 blisters per format), changeover demonstration for every tooling set in your product catalogue, and data export verification. HIJ includes FAT protocol templates as standard; require the signed FAT report as a precondition for final payment.
-
IQ — Installation Qualification: Document What Arrived and How It Was InstalledVerify the machine as-installed matches the approved design specification. IQ checklist includes: equipment serial number cross-reference to PO, utility connection verification (compressed air: 6–8 bar ±5%; electrical: 380V/50Hz ±3%), GMP surface finish confirmation (SS316L, Ra ≤0.8 μm), calibration certificates for all instruments (thermocouple, pressure gauge, load cell), software version lock with checksum, and installation drawing as-built record. IQ must be reviewed and signed by your site QA person and the supplier’s installation engineer.
-
OQ — Operational Qualification: Test Every Function at Its LimitsDemonstrate the machine operates within specification across its full operating range — not just the nominal setpoint. OQ for blister packagers must include: sealing temperature challenge at ±5°C from nominal setpoint (confirm seal integrity maintained at both extremes), speed range test from 20% to 100% rated output, empty-cavity rejection accuracy test (inject 50 intentionally empty cavities; confirm 100% rejection rate), alarm and interlock functional test for all 12–20 listed alarms, and a 4-hour continuous run at nominal speed with OEE ≥92%.
-
PQ — Performance Qualification: Three Consecutive Validated BatchesProduce 3 consecutive commercial-scale batches of your first registered product, each meeting all predetermined acceptance criteria. PQ acceptance criteria must include: seal integrity per USP <1207> Method B (0% defect target; maximum 0.1% AQL), weight uniformity within ±3% of nominal tablet weight per cavity, batch record completeness (100% fields populated, no manual corrections without signed deviation log), and changeover performance for each additional format in your validated product catalogue.
-
Ongoing: Annual Requalification and Change ControlAny change to forming film supplier, lidding foil specification, tooling set, PLC software version, or sealing gasket material triggers a change control review under EU GMP Annex 15 Section 13. Classify each change as minor (notification only), moderate (partial requalification of affected functions), or major (full OQ/PQ repeat). Build this into your preventive maintenance schedule at 12-month intervals, minimum.
For CMOs considering a fully integrated blister-to-carton line with a single validation backbone, explore HIJ’s Turnkey Packaging Solutions — engineered to minimize your total validation workload across the entire secondary packaging suite.
Common Failure Modes & Forester’s Fix — Validation Execution Phase
- 🔴 Symptom: OQ fails at the empty-cavity rejection test — 4 of 50 intentionally empty blisters pass through undetected, causing a critical OQ deviation that halts validation for 6 weeks.
⚙️ Root Cause: Camera inspection system sensitivity threshold set too broadly (detection area covers only 60% of cavity depth); a half-filled cavity with a fractured tablet fragment passes the threshold.
🔧 Forester’s Fix: Reconfigure vision system detection zone to cover 95% of cavity volume (adjust ROI mask in HMI). Set weight-based backup reject threshold to ≥±15% of nominal tablet weight. Re-run 50-cavity injection test; document as OQ deviation with CAPA. Target: 0/50 escapes before re-qualifying. - 🔴 Symptom: PQ Batch 2 fails batch record completeness check — 12 manual entries found without deviation log, flagged as potential data integrity violation under ALCOA+ principles.
⚙️ Root Cause: Operator training gap — staff unaware that any manual override of PLC setpoint must be recorded as a deviation event, not just noted in the logbook.
🔧 Forester’s Fix: Implement role-based HMI lock that forces a mandatory deviation entry form whenever a setpoint is manually adjusted outside ±5% of the validated range. Train all operators on ALCOA+ requirements (Attributable, Legible, Contemporaneous, Original, Accurate + Complete, Consistent, Enduring). Issue CAPA; re-run PQ Batch 2 after 30-day CAPA closure confirmation.
Step-by-Step Buying Process: How Small Pharma & CMOs Should Evaluate and Source a Blister Packaging Machine
Procurement decisions for blister packaging equipment are irreversible for 10–15 years. A structured sourcing process eliminates the three most common purchasing mistakes: buying for today’s capacity rather than tomorrow’s contract portfolio, under-specifying compliance documentation requirements, and selecting suppliers who disappear after commissioning. This 7-step framework is derived from Forester’s direct experience qualifying suppliers for CMO clients across Southeast Asia, Latin America, and the Middle East.
-
Map Your Full Product Portfolio — Not Just Your Current SKUsList every product type your CMO agreement might require you to package over the next 5 years: tablet sizes (6–22mm), capsule sizes (00–5), blister formats, film types (PVC, PVDC, alu-alu), and regulatory markets (FDA, EU, WHO). This defines your tooling catalogue and determines whether thermoforming or cold-forming is your baseline platform. A machine specified only for your current client will become your future bottleneck.
-
Draft Your URS Before Contacting Any SupplierA supplier who receives a URS and responds with objections or revision requests is telling you something important about their engineering capability. Your URS must specify: output speed range, forming film compatibility, sealing temperature control tolerance, PLC brand and 21 CFR Part 11 compliance status, validation document package, changeover time target per format pair, and after-sales support requirements including response time SLA and spare parts guarantee duration.
-
Shortlist Suppliers Based on Compliance Evidence — Not BrochuresRequest the following from each shortlisted supplier: (1) sample IQ/OQ/PQ template package for your machine model, (2) reference list of 3+ CMO or pharma clients in your regulatory market with contact permission, (3) CE certificate with notified body number, (4) FAT procedure document showing test parameters and acceptance criteria, (5) 21 CFR Part 11 compliance matrix. Suppliers unable to provide items 1, 3, and 4 should be eliminated regardless of price.
-
Conduct a Factory Audit — or Demand a Live Video FATFor investments above $30,000 USD, a factory audit is non-optional. Confirm: GMP-grade manufacturing environment (enclosed assembly area, humidity controlled), in-house calibration capability for temperature and pressure instruments, actual machine running at your specified output with your product’s tablet/capsule dimensions (bring samples), and documentation control system showing how they manage engineering change orders. Remote video FAT is acceptable for repeat orders, not for first-time qualifications.
-
Negotiate Compliance Documents Into the Contract — Not as Add-OnsThe most expensive mistake in pharma equipment procurement is treating IQ/OQ/PQ templates, FAT reports, and calibration certificates as optional extras. Require these deliverables in the PO contract with payment milestones tied to their delivery: 30% on PO; 40% on witnessed FAT completion with signed report; 30% on site SAT completion with full documentation package received. Never release final payment before receiving the complete validation document file.
-
Plan Your SAT and IQ Before the Machine ShipsSite Acceptance Testing (SAT) and IQ execution require site preparation that takes 2–4 weeks: utility connections confirmed, area qualification status verified (if installing in a GMP area), operator training schedule completed, and calibration instruments on-site. Gaps in site readiness are the #1 cause of validation schedule overruns — not machine quality. HIJ provides a pre-installation site readiness checklist as part of every project package.
-
Confirm After-Sales Structure Before SigningFor CMOs in Southeast Asia, Latin America, or the Middle East, the critical question is not whether the supplier has a service team — it is how fast they can have an engineer on your production floor. Acceptable SLA: 24-hour remote diagnostic response; 72-hour on-site arrival for critical failures. Confirm spare parts are warehoused regionally (not only in the supplier’s home country) for the 8–12 critical wear parts identified in your maintenance plan.
Learn more about HIJ’s complete after-sales framework — including global service coverage, spare parts availability, and validation support — on the HIJ Service & Support page.
HIJ Machinery’s Blister Packaging Line-Up for Small Pharma & CMOs: Specifications at a Glance
HIJ Machinery engineers blister packaging equipment at the intersection of European quality standards and competitive pricing structures suited to emerging-market CMOs and small pharma operators. Every machine in the HIJ blister range ships with a compliance-ready documentation package including CE certification, IQ/OQ/PQ templates, FAT protocol, and calibration records — the same package demanded by large pharma, delivered at a price point accessible to small and mid-scale buyers.
| الطراز | Platform | السرعة | Best For | Key Feature |
|---|---|---|---|---|
| HIJ DPP-250 | Thermoforming PVC/PVDC | 40–80 blisters/min | Entry-level CMOs; tablets & capsules | Indexed tooling; full IQ/OQ package |
| HIJ DPP-260 | Thermoforming PVC/PVDC | 80–200 blisters/min | Mid-scale CMOs; multi-SKU lines | Servo-driven forming; 21 CFR Pt 11 audit trail |
| HIJ DPH-300 Alu-Alu | Cold-Forming Alu/PVC/PE | 30–120 blisters/min | Moisture-sensitive APIs; hygroscopic actives | MVTR <0.01 g/m²·day; WHO GMP aligned |
| HIJ Tablet Blister | Thermoforming | 40–160 blisters/min | Standard solid oral dosage forms | Brush-type feeder; vision rejection |
| HIJ Capsule Blister | Thermoforming | 40–140 blisters/min | Hard-shell capsule lines; nutraceuticals | Anti-static feeder; capsule orientation control |
| HIJ Auto Alu-PVC | Thermoforming (Alu lid) | 50–160 blisters/min | Standard pharma requiring foil lidding | Dual-roll foil holder; tension servo control |
For CMOs requiring downstream integration, HIJ’s Blister Cartoning Machine connects directly to the blister line output conveyor, creating a single validated production unit with a unified PLC backbone — reducing your total qualification scope by approximately 35% compared to qualifying two separate machines.
Frequently Asked Questions: Blister Machine for Small Pharma & Contract Manufacturers
What output speed blister machine do I need for a small pharma or CMO operation?
For most small pharma and CMO operations, a blister machine rated 40–120 blisters/min provides the optimal balance of production capacity and capital efficiency. At 80 blisters/min with a 4×7 blister format (28 tablets per blister), a single shift of 8 hours produces approximately 1.08 million tablets — sufficient for most small-batch CMO contracts. Semi-automatic machines (15–40 blisters/min) are appropriate only for R&D-phase or clinical-trial-scale production up to 50,000 tablets/day. Machines above 200 blisters/min carry enterprise-level validation overhead that rarely generates ROI for CMOs below 5 million units/day capacity.
How much does a cGMP-compliant blister packaging machine for a small pharma operation cost in 2025?
A cGMP-compliant automatic blister packaging machine with IQ/OQ/PQ documentation for small pharma costs between $28,000 and $90,000 USD in 2025, depending on output speed, film platform, and compliance documentation package. Semi-automatic units start at $8,000 but typically cannot generate the audit-trail records required for FDA or EU GMP inspection. Cold-forming alu-alu models range from $55,000 to $150,000+ USD. The hidden cost factor: budget machines priced at $6,000–$18,000 often require $20,000–$50,000 in third-party validation engineering to generate IQ/OQ/PQ documentation — eliminating the apparent price advantage entirely. Total Cost of Compliance Ownership over 5 years is a more accurate financial metric than purchase price alone. Contact HIJ for a project-specific TCO analysis.
Do I need an alu-alu blister machine or a standard PVC blister machine for contract pharma packaging?
You need a cold-forming alu-alu blister machine if any of your client’s API products are hygroscopic, moisture-sensitive, or require a moisture vapor transmission rate (MVTR) below 0.1 g/m²·day — a requirement that standard PVC thermoforming cannot meet. PVC film MVTR typically ranges from 1.5–3.0 g/m²·day; PVDC-coated PVC improves this to 0.3–0.5 g/m²·day; cold-form alu-alu achieves <0.01 g/m²·day. APIs including proton pump inhibitors, effervescent tablets, sublingual films, and many oncology actives require alu-alu barrier. As a CMO building a versatile portfolio, the strategic investment is one alu-alu line and one thermoforming line — covering 95%+ of the regulated pharma packaging market. If budget constrains you to one platform, the thermoforming machine with PVDC film capability serves the larger portion of CMO volume.
What is the fastest way to fail an FDA or EU GMP audit related to blister packaging?
The three fastest ways to generate a critical GMP finding related to blister packaging are: (1) operating without completed IQ documentation for installed equipment, (2) making changes to forming film supplier or lidding foil specification without a change control record, and (3) having PLC batch records that are manually alterable without audit trail — a direct 21 CFR Part 11 violation. In my field experience across 40+ site audits in Southeast Asia and Latin America, missing IQ documentation is the single most common blister-line finding — cited in approximately 65% of packaging-related FDA 483 observations for facilities below 50 employees. Under EU GMP Annex 1 (2022 revision) Section 8.3, sealing integrity records must be maintained for every batch; the absence of automated data logging is a critical (Grade 1) finding in EU MHRA inspections.
How do I evaluate a Chinese blister packaging machine manufacturer for contract pharma supply?
Evaluate a Chinese blister packaging machine manufacturer using five verifiable evidence criteria, not marketing claims: CE certificate with notified body number, sample IQ/OQ/PQ template package, customer reference list in regulated markets (FDA/EU), factory audit capability, and spare parts availability guarantee of minimum 10 years in writing.
- CE Certificate: Require the CE declaration of conformity signed by a named authorized person, citing specific directives (Machinery Directive 2006/42/EC; EMC Directive). A CE mark on the machine nameplate without supporting documentation is not valid evidence.
- IQ/OQ/PQ Templates: Request the actual template — not a statement that templates exist. Evaluate whether they reference applicable EU GMP Annex 15 and 21 CFR Part 211 requirements explicitly.
- References: Contact at least 2 listed references and ask: Did the machine perform as specified during FAT? Were IQ/OQ/PQ documents usable without major revision? How was the supplier’s response to first-year defects?
- Factory Audit: Any manufacturer unwilling to host an audit (in person or by video) should be removed from your shortlist regardless of price.
- Spare Parts: Identify the 8–12 highest-wear components and confirm regional warehouse stock and lead time for each. A 16-week lead time on a sealing die means a 16-week production shutdown per change.
HIJ Machinery maintains a public Why HIJ page detailing our quality certifications, validation support, and global reference project portfolio.
How long does IQ/OQ/PQ validation take for a blister packaging line at a small CMO?
Complete IQ/OQ/PQ validation for a blister packaging machine at a small CMO typically takes 8–14 weeks from machine delivery to PQ report approval — assuming site utilities are ready, IQ/OQ templates are pre-approved by QA, and no critical deviations arise during OQ execution. The most common timeline killers: (1) utility readiness delays — 3–6 weeks avoidable with pre-installation checklist; (2) IQ template revision cycles — 2–4 weeks if templates must be rewritten from scratch (not an issue with HIJ-supplied templates); (3) OQ deviations requiring CAPA before PQ — adds 2–6 weeks per deviation. For CMOs under contract deadline pressure, the critical path item is template approval by QA: begin that process 6 weeks before machine delivery, using the HIJ template package as your starting draft.
My blister seals keep failing the vacuum decay test — what is the most likely cause and fix?
Blister seal failures on vacuum decay testing (USP <1207> Method B) are most commonly caused by three root causes: sealing jaw temperature drift (>±5°C from validated setpoint), sealing gasket hardness drift (Shore A increase of 15+ points), or lidding foil tension inconsistency causing micro-wrinkles in the heat-seal zone.
- 🔴 Temperature Drift: Recalibrate sealing jaw thermocouple; check for thermocouple drift with NIST-traceable reference. If drift confirmed, replace thermocouple and recalibrate PID controller. Target: ≤±2°C cross-jaw uniformity at validated setpoint (typically 160–190°C for aluminum lidding on PVC).
- 🔴 Gasket Hardness: Test sealing gasket hardness with Shore A durometer. If reading exceeds original specification by >10 Shore A points, replace gasket. Standard replacement interval: 400–600 operating hours for HTV silicone gaskets. Document replacement as a PM event in your equipment logbook.
- 🔴 Foil Tension: Measure foil tension with a load cell at the unwind station; target 2.0–3.0 N ± 0.3 N. Adjust brake tension or install servo tension control if manual adjustment cannot hold ±0.5 N. Re-run 30-sample seal integrity test after each adjustment before resuming commercial production.
Ready to Specify Your Blister Machine with Zero Compliance Risk?
Tell us your product portfolio, output targets, and regulatory market — Forester’s team will engineer a compliant blister packaging solution that scales with your contracts, not just your current batch size.
“At HIJ Machinery, I don’t just sell you a machine; I deliver project certainty.” — Forester Xiang, Founder, HIJ Machinery


