You’ve just received a blister packaging machine quotation. The spec sheet reads 200 blisters per minute. Your procurement team is pleased. Your production planning team builds a capacity model around that number. And then, six months after installation, your actual output hovers stubbornly at 110–130 blisters per minute — and your line manager has no idea why.
This gap between the number printed in a catalog and the number that actually appears on your shift report is not a malfunction. It is the industry’s most expensive and least-discussed performance illusion. Whether you’re specifying a new pharmaceutical blister packaging machine for a cGMP facility, or scaling up a nutraceutical line for export markets, understanding how to calculate real blister packaging machine output speed — not catalog numbers — is the single most important capacity-planning skill your engineering team can develop.

Founder, HIJ Machinery · 20 Years in the Field
🔥 A 20-Year Engineer’s Reality Check on Blister Machine Output
- The Direct Answer
Real blister packaging machine output speed is never the maximum mechanical speed stated in brochures. It is calculated by multiplying actual cycles per minute (CPM) by blisters per punch stroke, then applying a realistic Overall Equipment Effectiveness (OEE) rate — which, in well-managed cGMP facilities, sits between 75% and 82%. Any vendor who quotes you a “nameplate speed” without specifying OEE assumptions is selling you a fantasy. - The Field Experience
“In my 20 years conducting SAT and commissioning across Southeast Asia and South America, I have seen buyers paralysed by theoretical maximums more times than I can count. I recall a mid-sized pharmaceutical manufacturer in Indonesia who purchased a thermoforming blister line rated at 180 CPM. During SAT, running standard PVC/Alu with round tablets, it hit 172 CPM — respectable. But their actual product was a hygroscopic capsule in a 4×4 Alu-Alu format. The moment we switched to the real product, deep cold-form draw depth immediately forced the mechanical speed down to 120 CPM. Then, their downstream cartoner — sourced from a different supplier with a proprietary PLC protocol — couldn’t synchronise its infeed timing at that speed. The result: 20–25% waste rate from jamming and misalignment. Their real, sustainable output was 89 CPM. They had planned their entire SKU launch around 180.” - The Hidden Trap
Catalog speeds are always benchmarked on the most favourable conditions: shallow-draw PVC/Alu forming, round tablets with excellent flow characteristics, zero leaflet insertion, and a standalone machine with no downstream synchronisation pressure. The hidden trap is that no one in a competitive sales process volunteers this information. Furthermore, OEE erosion from multi-vendor line integration — where a blister machine, cartoner, and case packer from three different manufacturers must synchronise via Modbus, Ethernet/IP, or analogue triggers — can silently destroy 30–40% of your theoretical capacity before a single production batch has been validated.
The Master Formula: How to Calculate Real Blister Packaging Machine Output Speed
Before evaluating any supplier’s specification, your engineering team must be fluent in a three-layer calculation model. Ignore any one layer and your capacity planning will be wrong by a margin large enough to miss a product launch.
Real Output (blisters/min) = CPM × BPP × OEE%CPM = Actual Cycles Per Minute (material/format-adjusted, not nameplate)
BPP = Blisters Per Punch (number of blister cavities formed per single index stroke)
OEE% = Overall Equipment Effectiveness (Availability × Performance × Quality rate)
Layer 1 — Cycles Per Minute (CPM): This is the most misrepresented number in blister machine specifications. Nameplate CPM is tested under ideal conditions: shallow PVC forming depth (typically 5–8 mm), fast-moving round tablets, no upstream feeding variability. In real production, CPM degrades according to material physics. A cold-form Alu-Alu process forming at 12–16 mm draw depth with foil thicknesses of 45–60 µm applies significant back-pressure on the forming station’s mechanical indexer, mandating a 15–25% speed reduction to maintain seal integrity and prevent pinhole defects. A servo-driven machine can compensate partially by optimising dwell timing at the forming station, but physics cannot be legislated away.
Layer 2 — Blisters Per Punch (BPP): This multiplier is often overlooked in capacity conversations, yet it is the single highest-leverage variable in your output equation. A machine running at 100 CPM with a 2×5 (10-cavity) tooling produces 1,000 blisters per minute. The same machine with a 4×5 (20-cavity) tool produces 2,000. However, wider tooling increases forming tonnage requirements, demands precise temperature uniformity across a larger heat plate, and imposes tighter tolerances on the film web-guiding system. Specifying maximum BPP without verifying the machine’s servo torque capacity and heat-plate uniformity (±2°C tolerance is the standard for pharmaceutical PVC/PVDC forming) is a common engineering oversight.
Layer 3 — OEE Decomposition: OEE is the most honest number in any packaging line assessment. It is the product of three sub-metrics: Availability (unplanned downtime as a percentage of scheduled production time), Performance Rate (actual speed vs. ideal speed), and Quality Rate (good units as a percentage of total units started). A world-class pharmaceutical blister line achieves OEE of 85%+. A realistic benchmark for a well-maintained mid-tier line in a regulated facility is 75–80%. A poorly integrated multi-vendor line with recurring jam events and cross-communication faults commonly drops to 55–65% — a figure that will destroy your production economics within two fiscal quarters.

Fig. 1 — HIJ servo motor control system: the primary hardware variable governing real CPM accuracy under format-specific forming loads.
Format Variables That Silently Destroy Your Output Speed
Once you have the formula, the next critical discipline is understanding that every format change is a new speed calculation. Many procurement teams finalise a machine purchase based on one product trial and then discover that the remaining 60% of their SKU portfolio runs at radically different speeds. Here is the engineering reality of the four most common format variables:
| Format Variable | Catalog Condition | Real-World Condition | Typical Speed Impact |
|---|---|---|---|
| Forming Material | PVC 250 µm / Alu lidding | Alu-Alu cold-form (45 µm + 60 µm) | −15% to −25% CPM |
| Forming Depth | 5–8 mm (tablets) | 12–16 mm (deep-draw capsules) | −10% to −20% CPM |
| Product Shape / Flow | Round tablet, excellent flow | Oblong / hygroscopic capsule, sticky | −5% to −15% CPM + higher waste rate |
| Leaflet Insertion | No leaflet (standalone test) | Folded leaflet, 4-fold, in-line | −5% to −10% line rate |
| BPP Tooling Width | 2×5 standard tooling | 4×5 wide-web tooling | +100% output IF heat plate uniform |
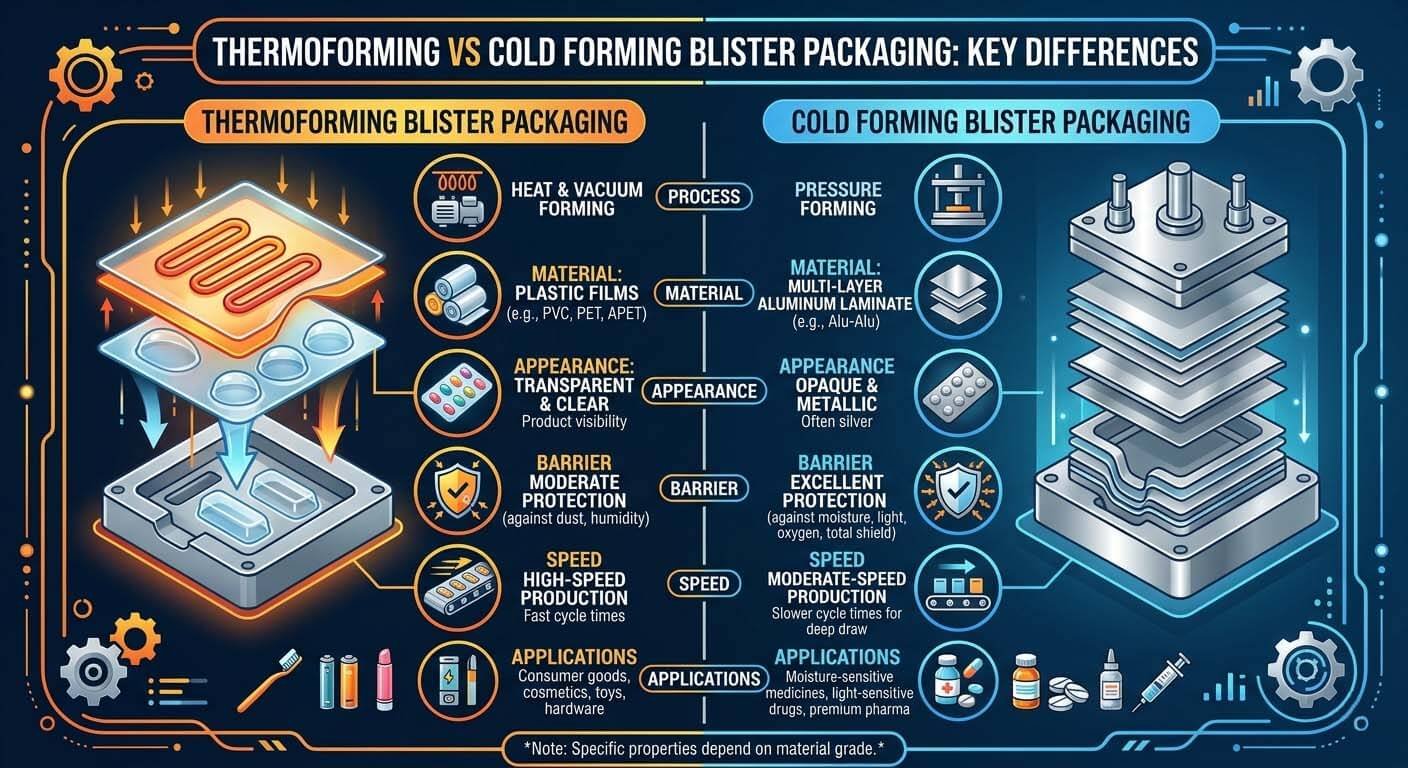
Fig. 2 — Thermoforming (PVC/Alu) vs. Cold-Form (Alu-Alu): the single biggest format variable driving real-world CPM deviation from catalog specifications.
OEE Deep Dive: Where Your 30% Capacity Disappears Every Shift
Most production engineers understand OEE as a concept. Far fewer have done the arithmetic on exactly where OEE losses accumulate on a blister line. Based on HIJ’s SAT commissioning data from over 100 pharmaceutical and nutraceutical facilities globally, here is the breakdown of where a typical mid-tier installation loses performance:
| OEE Loss Category | Root Cause | Typical Impact | HIJ Standard Mitigation |
|---|---|---|---|
| Planned Downtime | Mould changeovers, format changeovers | 4–8% availability loss per 8-hr shift | Quick-release tooling, pre-set servo recipes |
| Unplanned Downtime | Film web breaks, feeder jams, PLC faults | 3–12% availability loss | Predictive tension monitoring, Siemens / Allen-Bradley PLC with full fault logging |
| Speed Loss | Running below ideal CPM due to material issues | 10–20% performance rate loss | Format-specific servo speed recipes locked at FAT |
| Minor Stops | Tablet misfeeds, blister rejection events under 1 min | 5–10% performance rate loss | Integrated vision inspection + auto-rejection with no line stop |
| Quality Rejects | Seal integrity failures, empty cavities | 1–5% quality rate loss | In-line seal pressure monitoring, camera inspection per blister card |
| Line Sync Faults | Multi-vendor PLC desync with cartoner / case packer | Up to 40% OEE collapse in worst cases | Unified Turnkey PLC protocol, single SCADA point of control |
The most operationally devastating category in the table above is the final one: line synchronisation faults. When your blister machine, cartoner, and case packer originate from different manufacturers, each with its own PLC firmware, communication protocol (Modbus RTU vs. EtherNet/IP vs. PROFIBUS), and reject-signal handshaking logic, the integration burden falls entirely on your in-house engineering team. In practice, this results in microsecond timing misalignments at the transfer conveyor between blister machine and cartoner infeed, leading to blister card misorientation, cartoner pocket jams, and cascading line stops. I have witnessed complete production runs aborted in regulated facilities because the multi-vendor line could not maintain synchronisation for more than 40 continuous minutes during Process Performance Qualification (PPQ).
Multi-Vendor Patchwork vs. HIJ Turnkey: An Engineering Cost-Benefit Analysis
The decision to source a complete blister packaging line from multiple vendors vs. a single turnkey integrator is one of the most consequential capital expenditure decisions a pharmaceutical or nutraceutical manufacturer makes. The arguments for multi-vendor sourcing are intuitive: competitive pricing on individual units, leverage over suppliers, familiarity with specific brands. The arguments for turnkey integration are engineering and operational — and they compound over time.
- 3–5 separate FAT events at different factories
- No single point of integration responsibility
- PLC protocol mismatches require custom middleware
- OEE benchmarking impossible across the line
- Warranty disputes when issues cross machine boundaries
- SAT commissioning delays of 4–8 additional weeks
- SCADA integration requires third-party consultant
- Spare parts from multiple warehouses, multiple lead times
- Single FAT covers blister machine + cartoner + case packer
- Unified Siemens or Allen-Bradley PLC architecture
- Pre-validated inter-machine communication protocols
- Guaranteed OEE continuous run clause in FAT protocol
- One URS document, one validation package (IQ/OQ/PQ)
- Single warranty scope, zero integration disputes
- Real-time SCADA with centralised OEE dashboard
- Global spare parts inventory, 24-hr dispatch commitment
At HIJ, our turnkey packaging line solutions are engineered from a single philosophical premise: the only speed that matters is the speed at which validated, releasable product exits your case packer — not the theoretical maximum of any individual machine in the chain. Every HIJ turnkey line undergoes a mandatory 4-hour OEE continuous run at FAT, using your actual product format, your actual forming materials, and your actual downstream carton specifications. The OEE result is documented, signed, and becomes a contractual performance guarantee.

Fig. 3 — HIJ turnkey integrated blister packing line: blister machine, cartoner, and case packer on a unified PLC architecture — the only configuration that guarantees real output speed.
Demanding Real Output Proof: The FAT/SAT Protocol You Must Enforce
Knowing the formula is not enough. The formula must be contractually enforced through a rigorous FAT (Factory Acceptance Test) and SAT (Site Acceptance Test) protocol. Here is the minimum standard your quality and engineering team should demand for any blister packaging machine procurement above USD 150,000:
FAT — Factory Acceptance Test requirements: The FAT must be conducted with your actual forming film (same material grade, supplier, and roll batch), your actual product (or a validated placebo of identical geometry and bulk density), and your target BPP tooling. The test must include a minimum continuous run of 2 hours at target CPM to generate statistically valid OEE data. Seal integrity must be verified by dye-penetration test (per ISO 11607) and peel-force measurement. Camera inspection rejection accuracy must be validated against a defined defect library. All servo recipe parameters (forming temperature, sealing pressure, index speed, and dwell time) must be documented in the Validated Equipment Parameter Record (VEPR) and locked under user-level access control.
SAT — Site Acceptance Test requirements: Post-installation, the SAT must re-run the same 2-hour continuous OEE test, now including the live cartoner and case packer integration. Line synchronisation must be verified at three speed setpoints: 60% of target CPM, 85% of target CPM, and 100% of target CPM. Any reject event during the SAT must be captured by the PLC fault log, and the mean time to recovery (MTTR) must be ≤90 seconds. The complete SAT dataset forms the foundation of your OQ (Operational Qualification) protocol under 21 CFR Part 211.

Fig. 4 — Seal integrity verification during FAT: a non-negotiable quality gate that confirms real CPM is sustainable without compromising sealing pressure and blister card integrity.
Quick-Change Tooling: The OEE Multiplier Most Engineers Undervalue
One of the most operationally significant, yet consistently undervalued, contributors to real blister packaging machine output speed is changeover time. Every format changeover — changing BPP tooling, forming depth, or film web width — takes the machine offline. On a high-SKU pharmaceutical line running 8–12 formats per week, changeover-driven availability loss can represent 12–18% of total scheduled production time.
The engineering solution is quick-change tooling architecture. HIJ’s DPP-260 automatic blister packing machine incorporates a tool-free, indexed locating plate system for the forming and sealing dies. Format changeover — including mould swap, PLC recipe recall, and web re-threading — is achievable in 18–22 minutes by a single trained operator, against an industry average of 45–75 minutes for conventional bolt-pattern tooling. When this changeover time advantage is annualised across a 5-day production week, the OEE gain from quick-change tooling alone can recover 200–350 additional production hours per year — the equivalent of 4–7 additional production weeks at no capital cost.

Fig. 5 — HIJ quick-change mould system: indexed locating plates allow single-operator format changeover in under 22 minutes, recovering up to 350 production hours annually versus conventional tooling.
Pertanyaan yang Sering Diajukan
What is a realistic OEE target for a pharmaceutical blister packaging line in a cGMP facility?
A well-managed, single-vendor or turnkey-integrated pharmaceutical blister line operating under WHO GMP or FDA cGMP conditions should target an OEE of 75–82% as a sustainable production benchmark. World-class performers with high-volume, single-format lines can reach 85–88%. Anything below 70% sustained over a 90-day period indicates a systemic integration, maintenance, or process optimisation problem that requires root-cause investigation. It is critical to note that an OEE of 75% on a machine rated at 200 CPM means your real sustainable output is 150 CPM — a 25% capacity gap that will materialise as a missed production target within the first quarter of operations if not planned for at the URS stage.
How does Alu-Alu cold-form blister packaging affect machine speed compared to standard PVC/Alu thermoforming?
Cold-form Alu-Alu blister packaging — used for moisture-sensitive and light-sensitive drugs requiring maximum barrier protection — imposes a 15–25% mechanical speed penalty compared to PVC/Alu thermoforming. The reasons are multi-factorial: cold-form foil (typically a 45 µm OPA / 45 µm Al / 60 µm PVC laminate) requires significantly higher forming force (2–3× that of thermoplastic PVC), longer dwell time at the forming station to achieve consistent draw depth without pinhole defects, and tighter web tension control to prevent foil micro-cracking. On a HIJ Alu-Alu blister packing machine, servo forming-press parameters are pre-configured for each specific foil laminate specification in your URS, ensuring the speed reduction is minimised and documented during FAT rather than discovered during production.
Can I use a blister machine’s blisters-per-minute (BPM) catalog figure directly for production capacity planning?
No — and doing so is the single most common capacity-planning error in pharmaceutical packaging project management. The catalog BPM figure represents the machine’s theoretical maximum mechanical output under ideal test conditions, with zero OEE discount applied. For capacity planning, you must apply the full three-layer calculation: Real Output = CPM × BPP × OEE%, using your specific forming material, your actual product’s CPM-adjusted speed, and a conservative OEE of 75–78%. If a supplier cannot tell you what OEE assumption their catalog number is based on, that is a red flag. At HIJ, every quotation includes a format-specific capacity matrix that explicitly states the OEE assumption, the adjusted CPM for your product type, and the resulting real output per shift — in writing, before you sign a purchase order.
How does increasing Blisters Per Punch (BPP) tooling affect machine complexity and cost?
Increasing BPP (e.g., moving from a 2×5 to a 4×5 tooling configuration) doubles theoretical output without increasing CPM, making it the highest-leverage output multiplier available. However, wider tooling introduces several engineering constraints: heat plate temperature uniformity (PVC thermoforming requires ±2°C across the entire forming surface), servo forming-press tonnage (force required scales linearly with forming area — a machine undersized in press tonnage will compensate by slowing CPM, eliminating the BPP advantage), and web tension management (wider webs require proportionally more sophisticated dancer-roll tension control). HIJ’s standard tooling platform supports BPP configurations up to 4×8 on the DPP-260 high-output blister machine, with servo-controlled heat plates verified to ±1.5°C uniformity across the full forming width at FAT.
Conclusion: Delivering Project Certainty on Blister Output Speed
The gap between a catalog speed and a real, validated, sustainable production output is not a minor rounding error — it is routinely a 20–40% capacity shortfall that manifests as missed production targets, failed PPQ runs, regulatory scrutiny, and erosion of the financial model that justified your capital investment. The formula is not complicated: Real Output = CPM × BPP × OEE%. The discipline is applying it rigorously, with your actual materials, your actual products, and your actual downstream integration scenario — before you commit to a purchase order.
Define your URS with format-specific speed requirements. Demand a format-specific OEE continuous run at FAT. Require a single-point turnkey integration responsibility so that PLC synchronisation, SCADA communication, and inter-machine timing are guaranteed — not left as a post-installation negotiation between vendors who each claim the other is at fault.
At HIJ Machinery, I don’t just sell you a blister packaging machine — I deliver project certainty. That means a guaranteed, validated output number on your actual product, in writing, before you ship a single machine.
Stop Guessing Your Real Output. Let Our Engineers Calculate It For You.
Send us your product format, forming material, BPP requirement, and target output — and Forester’s team will return a format-specific capacity matrix with guaranteed OEE assumptions within 48 hours.

