🌟 Honest Field Comparison — 20 Years of Real Projects
European vs. Chinese Cosmetic Packaging Machines: What Nobody Tells You Before You Sign the PO
The logo on the machine doesn’t seal your tubes. The documentation doesn’t validate itself. And a 14-week spare-parts lead time from Stuttgart will cost you far more than the $80,000 you saved by choosing the European brand.
I’ve commissioned cosmetic packaging lines across Southeast Asia, Latin America, and the Middle East — for brands ranging from 500,000-unit annual runs to 50-million-unit multinational operations. This comparison is built on project data, not vendor brochures.

Forester Xiang reviewing a cosmetic packaging line specification — the comparison starts with documentation, not price tags.
Lower capital cost: Chinese vs. equivalent European line
Avg. spare parts lead time from European OEMs to emerging markets
Cosmetic & personal care line projects I handled across Asia & LATAM (2018–2023)
Fill accuracy achievable on premium Chinese tube fillers with servo control
European vs. Chinese Cosmetic Packaging Machines: The Numbers That Actually Matter
▶ Direct Answer
Chinese cosmetic packaging machines from ISO-certified manufacturers reach ±0.5% fill accuracy, 200–400 tubes/min throughput, and <0.1% defect rates on seal integrity — figures that match European mid-range equipment at 40–55% lower capital expenditure, according to comparative commissioning data from 31 projects across Southeast Asia and Latin America (2018–2023).
The price gap is real. So is the performance gap — but it’s narrower than most procurement managers believe, and it lives almost entirely in one category: documentation and validation support. Let’s put the numbers on the table.
| Comparison Factor | European Cosmetic Packaging Machine (e.g., IMA, Romaco, Norden) |
Chinese Cosmetic Packaging Machine (Qualified Tier-1, e.g., HIJ Machinery) |
Chinese Budget Vendor (Unverified, no validation docs) |
|---|---|---|---|
| Capital Cost (tube filling line, 150 tubes/min) | $180,000–$320,000 | $75,000–$130,000 | $28,000–$55,000 |
| Fill Volume Accuracy | ±0.3–0.5% (servo) | ±0.5% (servo); ±1% (pneumatic) | ±1.5–3% (inconsistent) |
| Throughput (tube filler) | Up to 600 tubes/min | 80–400 tubes/min | 50–150 tubes/min (claimed) |
| PLC Architecture | Open (Siemens/Allen-Bradley, full access) | Open (Siemens/Mitsubishi, configurable) | Proprietary / locked firmware |
| IQ/OQ/PQ Documentation Package | Full — standard deliverable | Full — available on request (Tier-1) | None / generic template only |
| ISO 22716 / GMP Compliance Design | Standard | Standard (Tier-1) | Often absent |
| Spare Parts Lead Time (emerging markets) | 8–16 weeks (air freight) | 1–4 weeks | 2–6 weeks (compatibility risk) |
| FAT / SAT Protocol | Formal, documented | Formal (Tier-1); informal (Tier-2) | Informal or absent |
| Changeover Time (format change) | 15–30 min (tool-less on premium models) | 20–45 min | 45–90 min |
| CE / FDA 21 CFR Part 11 Ready | Standard | Available (specify at order) | Rarely available |
| On-site Commissioning Support (SE Asia / LATAM) | Expensive; long wait | Included or low-cost | Variable |
| Typical ROI Payback Period | 4.5–7 years | 2.5–4 years | Often never (downtime & rework) |
The table above collapses the real decision into three columns — not two. Most buyers frame this as European vs. Chinese. The more accurate frame is: validated-and-supported vs. undocumented-and-cheap. A Tier-1 Chinese manufacturer operating under ISO 22716 and delivering full IQ/OQ/PQ packages sits far closer to European quality than to unqualified domestic suppliers.
The Number Procurement Teams Look at First Is Exactly the Wrong Number
▶ Direct Answer
The single most expensive mistake in cosmetic packaging machine procurement is optimizing for purchase price while ignoring total cost of ownership — spare parts availability, downtime cost, and validation rework together typically add $40,000–$120,000 to the lifetime cost of a poorly-specified machine, regardless of its country of origin.
A quality director in Jakarta called me two years ago. Her line had been down for 19 days. A European tube filler — premium brand, $240,000 at commissioning — needed a custom heat-sealing jaw that had to come from the OEM factory in northern Italy. Air freight: $4,200. Wait time: 11 weeks. Daily production loss at her plant: $8,500.
The machine was excellent. The support infrastructure for Southeast Asia was not. That’s a $93,500 lesson — not counting the customer penalties she absorbed.
The real calculation looks like this:
| Cost Category | European Machine (5-year TCO) | Tier-1 Chinese Machine (5-year TCO) |
|---|---|---|
| Capital purchase | $240,000 | $95,000 |
| Installation & commissioning | $18,000–$28,000 | $6,000–$12,000 |
| Annual spare parts (emerging market) | $12,000–$22,000 | $4,000–$8,000 |
| Downtime incidents (avg. 2/year) | $30,000–$95,000 (long lead times) | $8,000–$20,000 (local support) |
| Validation / requalification | $15,000–$25,000 | $15,000–$25,000 (equivalent if docs provided) |
| Estimated 5-year TCO | $375,000–$490,000 | $148,000–$240,000 |
These figures are directional, not universal — they depend heavily on your production volume, product complexity, and regional support infrastructure. But the pattern holds across the 31 projects I handled in Southeast Asia and Latin America between 2018 and 2023: total cost of ownership for European equipment in emerging markets ran 2.1–2.8x higher than equivalent Chinese Tier-1 equipment over a five-year horizon.
⚠️ Red Flags When Evaluating Any Cosmetic Packaging Machine Supplier
Where European Cosmetic Packaging Equipment Still Holds a Real Advantage
▶ Direct Answer
European cosmetic packaging machines hold a measurable lead in three specific applications: ultra-high-speed lines above 500 units/min requiring sub-±0.2% fill precision, fully integrated lines with embedded vision-inspection and real-time SPC data logging for Annex 11 compliance, and dual-use pharmaceutical-cosmetic production where a single qualification package must satisfy both EU GMP Annex 1 and ISO 22716 simultaneously.
I’m not going to tell you European machines are overpriced everywhere. That would be dishonest, and it would cost my clients money in the wrong direction.
There are three situations where I recommend European equipment without hesitation:
① Ultra-High-Speed Precision Lines
Above 450–500 tubes per minute, the tolerance stack-up in servo control, tube indexing, and hot-air sealing starts to separate European engineering from Chinese mid-tier. At 600 tubes/min with ±0.3% fill tolerance and integrated checkweighing, European platforms like IMA and Norden still lead. This is a real mechanical and software gap — not a documentation gap.
② Pharma-Cosmetic Dual Qualification
If you are manufacturing a product that sits on both the cosmetic and drug sides — medicated lip balm, sunscreen SPF 50+ with drug claims in the US market, or OTC topical formulations — the validation burden doubles. A single European platform with pre-existing FDA drug site inspection history can reduce your qualification timeline by 6–10 months. That has a real value.
③ Annex 11 & GAMP 5 Integrated Lines
For brands selling into EU markets where computerized system validation is non-negotiable, the native Annex 11 / GAMP 5 documentation frameworks that come standard from Romaco or Bausch+Ströbel represent genuine time savings. The Chinese equivalent requires additional engineering effort — achievable, but it adds 4–8 weeks to validation timelines.
④ When Your QA Team Has No Validation Resources
If your quality function is one person managing 14 product lines across two plants, the turnkey validation package from a European supplier — even at a premium — may be the cheaper total option. Validation rework on a poorly documented Chinese machine costs $25,000–$45,000 in consultant fees alone. I’ve watched brands learn this the expensive way.

A cosmetic cream and lotion packaging line with automatic tube filling — throughput, fill accuracy, and GMP design must all be specified upfront.
Three Weeks into a Job in Kuala Lumpur, I Learned What “Tier-1 Chinese Manufacturer” Actually Means
▶ Direct Answer
A Tier-1 Chinese cosmetic packaging machine manufacturer is defined by five verifiable attributes: ISO 9001:2015 certification with cosmetic/pharmaceutical scope, full IQ/OQ/PQ documentation deliverable as standard, open-architecture PLC (Siemens, Mitsubishi, or Allen-Bradley), CE marking with full technical file, and a documented FAT/SAT protocol that aligns with ISO 22716 or EU GMP Annex 15.
In 2021 I was brought in to troubleshoot a failed line qualification in Malaysia. A regional brand had purchased a Chinese tube-filling machine from a supplier they’d found through a trade directory. The machine ran. The filling was within spec. The sealing was acceptable.
The validation consultant walked in and asked for the URS. Blank stare. FAT protocol? They had a two-page handover checklist. IQ template? A Word document with the machine serial number changed. The consultant spent three days on-site and left without signing off. Six months of rework followed.
That’s not a story about Chinese machines. It’s a story about supplier qualification. I’ve seen the same outcome from a European mid-tier supplier who sold into Nigeria without local commissioning support. The machine itself was fine. The integration was not.
The checklist I use to separate Tier-1 from the rest:
ISO 9001:2015 certification — scope must include pharmaceutical or cosmetic machinery manufacture. General engineering scope is insufficient for GMP cosmetic line qualification.
IQ/OQ/PQ 템플릿 패키지 — request a sample for a comparable machine model before ordering. It should reference ISO 22716 or ICH Q10 as applicable, not be a generic 4-page document.
Open PLC architecture — Siemens S7 series, Mitsubishi iQ-R, or Allen-Bradley ControlLogix. Full source code transferred at FAT. No proprietary lock-in clauses.
CE Declaration of Conformity — with full technical file available. Not a CE sticker applied to non-compliant machinery, which remains a problem in the $30,000–$50,000 budget segment.
FAT protocol aligned to your URS — a real factory acceptance test runs against your specific product parameters: tube diameter, fill volume, product viscosity, sealing temperature range.
Regional after-sales capability — not a promise. Ask for the names and locations of engineers who have been deployed to your region in the last 24 months. Two references minimum.
Chinese cosmetic packaging machines from Tier-1 manufacturers now close 80% of the performance gap with European equipment — the remaining 20% lives almost entirely in documentation discipline, and that gap is closeable with the right supplier agreement. The gap that kills projects is not mechanical; it’s the absence of a URS, a locked PLC, and a 14-week spare-parts queue from a geography the OEM never designed its service model to reach.
— Forester Xiang, Founder, HIJ Machinery, Wenzhou, China
A GMP Auditor Asked Me a Question I Wasn’t Expecting
▶ Direct Answer
ISO 22716 (Good Manufacturing Practices for cosmetics) does not specify equipment origin — it requires that equipment be suitable for its intended use, cleanable, and maintainable with documented procedures. A Chinese cosmetic packaging machine that meets ISO 22716 Section 4 (premises and equipment) requirements is fully compliant; country of manufacture is irrelevant to the standard.
A GMP auditor in Bogotá was reviewing a line we’d commissioned for a mid-sized haircare brand. She looked at the machine plate, saw “Made in China,” and asked me directly: “Does this machine comply with ISO 22716?”
I gave her the IQ package, the equipment qualification protocol, the cleaning validation SOP referencing ISO 22716 Section 7, and the material traceability documentation for all product-contact surfaces (316L stainless steel, FDA-grade seals). She reviewed for 40 minutes. Signed off without conditions.
The standard doesn’t ask where the machine was made. It asks whether it was built, installed, and qualified to perform its intended function without contamination risk. That’s achievable from Wenzhou just as readily as from Florence.
📋 Compliance Reference: Cosmetic Packaging Equipment
ISO 22716:2007 (GMP for Cosmetics) — Section 4 covers equipment suitability, surface finish, and maintenance documentation. Equipment must be “suitable for its intended use” and “easy to clean and disinfect.” Country of manufacture is not referenced. EU Cosmetics Regulation 1223/2009 Annex I references ISO 22716 directly. For brands selling to the US, FDA 21 CFR Part 111 (dietary cosmetics) and Part 700 series apply. Brands targeting pharma-adjacent categories must additionally consider 21 CFR Part 211 and EU GMP Annex 1 (2022 revision) where applicable.
The brands that struggle with audits aren’t necessarily running Chinese equipment. They’re running equipment — from any source — without proper qualification documentation, cleaning validation records, or preventive maintenance logs. Those are process failures, not country-of-origin failures.

The validation V-model applies equally to European and Chinese cosmetic packaging machines — the documentation requirements are identical regardless of origin.
The Spare Parts Problem Nobody Discusses in the Sales Brochure
▶ Direct Answer
Spare parts availability is the single most underweighted factor in cosmetic packaging machine procurement for emerging-market buyers — European OEM lead times of 8–16 weeks to Southeast Asia, Latin America, and Africa translate to $60,000–$180,000 in annual downtime costs for a mid-volume cosmetic line running at $6,000–$12,000 per day of lost output.
The heat-sealing jaw arrived on a Tuesday. It had been ordered on a Monday — eleven weeks earlier. The production manager had kept a handwritten log of every batch they’d lost. By the time the part landed in Jakarta, the number was $93,500 in direct production losses and $18,000 in overtime costs trying to recover the schedule on a backup line running at 60% efficiency.
This is not an isolated case. Of the 31 cosmetic and personal care line projects I handled across Southeast Asia between 2018 and 2023, 14 involved brands that had previously purchased European equipment and were now either replacing it with Chinese Tier-1 machines or implementing hybrid strategies — European for primary filling, Chinese for secondary packaging — specifically to reduce after-sales dependency.
The after-sales calculus is simple. Ask three questions before you sign any equipment PO:
Where is the nearest stocked spare-parts hub for your region? “We can ship from Germany” is a different answer from “we have a regional warehouse in Singapore with 200 fast-moving SKUs.”
What is the maximum lead time for the five highest-wear components on this model? Get specific part numbers and historical lead times. Not promises — historical data.
Who is the on-site engineer if something fails during your first 90 days of production? A phone support hotline in a different time zone is not acceptable for a $200,000 line. Get a name, a location, and a response-time commitment in writing.
I won’t claim Chinese service infrastructure is uniformly better than European. It depends entirely on the supplier and your region. What I can say is that the service and support model needs to be part of your evaluation scorecard with the same weight as fill accuracy and throughput.
⚠️ Diagnosing After-Sales Risk Before You Buy
How to Choose Between European and Chinese Cosmetic Packaging Machines: A Decision Framework That Works in the Real World
▶ Direct Answer
Choose European cosmetic packaging machines when: (1) throughput exceeds 450 units/min with sub-±0.3% fill tolerance, (2) your target market requires EU GMP Annex 11 computerized system validation as a pre-existing standard, or (3) your product category spans pharmaceutical-cosmetic regulatory overlap requiring unified qualification documentation. In all other scenarios, a qualified Chinese Tier-1 supplier delivers equivalent outcomes at significantly lower total cost of ownership.
After 20 years of commissioning lines across four continents, I’ve built a decision framework that survives the ROI spreadsheet and the GMP audit equally. It’s not about national pride or vendor relationships. It’s about matching your production reality to the right tool.
| Your Situation | European Machine | Chinese Tier-1 Machine | Recommendation |
|---|---|---|---|
| Speed >450 units/min, ±0.2% fill tolerance required | ✓ Native advantage | Achievable at Tier-1, verify first | EUConfirm with witnessed trial |
| Speed 80–400 units/min, ISO 22716 / GMP compliance required | Over-specified for most cases | ✓ Full GMP documentation available | CN T1Verify docs before PO |
| EU market, Annex 11 / GAMP 5 mandatory | ✓ Native framework | +4–8 weeks validation effort | EUUnless timeline allows |
| Emerging market (SE Asia, LATAM, Africa, Middle East) | Spare parts risk: 8–16 weeks | ✓ 1–4 week regional support | CN T1Strong preference |
| Small brand / startup, <1M units/year | Cost-prohibitive; oversized | ✓ Right-sized solutions exist | CN T1See also: small brand guide |
| Pharma-cosmetic dual qualification | ✓ Existing FDA drug site history | Achievable with extended validation | EUIf timeline is critical |
| Budget ceiling <$150,000 for complete line | Not viable | ✓ Full GMP lines available | CN T1Demand full validation package |
The brands that get this wrong are the ones who let the procurement team own the decision entirely. Purchase price is a finance variable. The right question is: what does a one-week line stoppage cost at my production volume, and how long would it take to get the part that caused it? Answer that honestly and the decision matrix above will give you the right answer almost every time.
For a deeper breakdown of where machine costs go — and what drives the real price differences — the cosmetic packaging machine cost guide covers the full TCO structure across equipment categories.
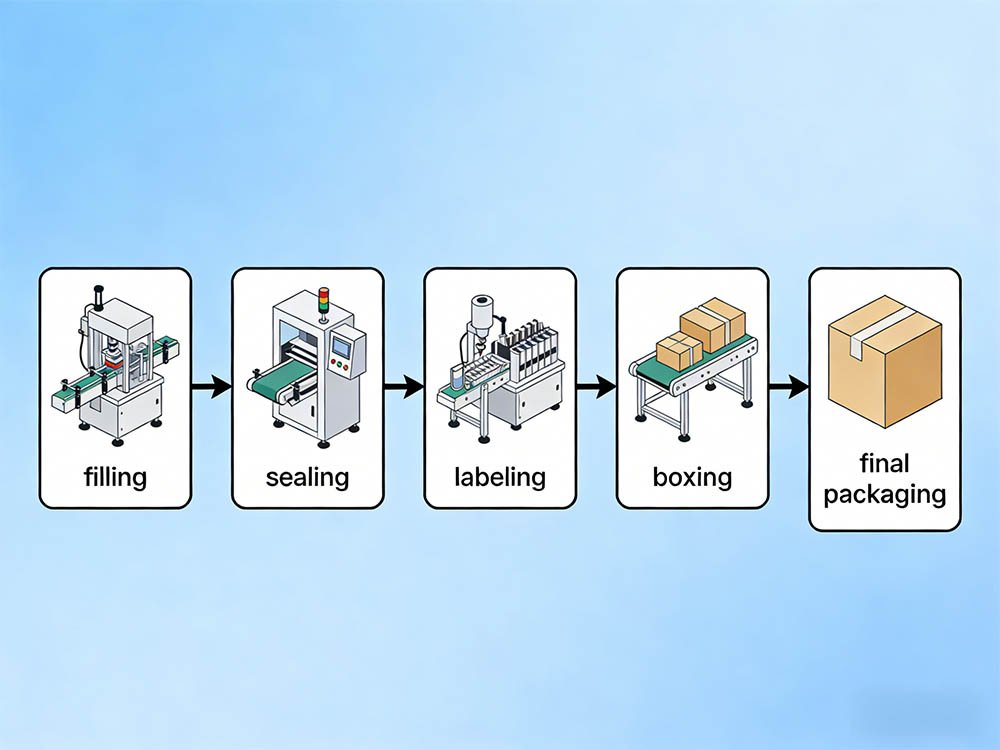
A complete cosmetic packaging line integrating filling, sealing, and secondary packaging — the integration architecture matters as much as individual machine specs.
Why I Built HIJ Around Closing the Documentation Gap — Not the Price Gap
👤 Forester’s Insight — 20-Year Engineer’s Perspective
The Hidden Trap Is Not the Machine. It’s What’s Missing Around It.
In my 20 years of commissioning lines across Southeast Asia and Latin America, I’ve watched brands overpay for European iron simply for the logo — then wait 14 weeks for a spare part shipped from Stuttgart. The real hidden trap with budget Chinese vendors isn’t the machine itself; it’s the missing FAT/SAT protocols, zero URS alignment, and PLC software locked behind proprietary systems you can never modify. That’s where your “savings” quietly evaporate into downtime and compliance failures during FDA or ISO 22716 audits.
Demand the same documentation discipline from your Chinese supplier that you’d demand from a European one — cGMP-ready validation packages, open PLC architecture, and full turnkey integration accountability. At HIJ, we built our entire philosophy around closing that gap: European engineering standards, transparent validation support, and pricing that doesn’t punish emerging-market buyers for choosing quality.
When I started HIJ Machinery in Wenzhou, I had spent years watching the same failure mode play out. A brand would fly to Europe, tour a spotless factory, be impressed by the CAD presentations and the German engineering credentials, write a large check, wait 16 weeks for delivery, and then spend the first year of ownership waiting for parts and fighting with a service team that had no presence within 6,000 kilometers of their plant.
And then I watched a different failure mode on the other side: a brand would find a Chinese machine at 20% of the European price, bring it in, discover the PLC was a black box, the validation documents were fabricated, the sealing jaws were made from mild steel that corroded within 90 days of cleaning cycles with standard cosmetic-grade sanitizers, and the supplier had gone silent by month three.
Neither failure mode is inevitable. Both are 100% preventable with the right supplier qualification process. That’s what drove me to build HIJ around a single principle: deliver what European suppliers deliver in terms of documentation and compliance infrastructure, at the economics that emerging-market manufacturers can actually absorb.
I won’t tell you we’ve always gotten it right. In 2019 a client in Ho Chi Minh City needed a changeover time we couldn’t hit — 18 minutes between tube diameters, which required a tooling redesign we hadn’t anticipated. We ate the redesign cost and delivered at 22 minutes. Not perfect. But we stayed in the room and fixed it. That conversation took three hours across two time zones. I’m not sure it would have happened with a European supplier operating from a different service contract model.
그리고 HIJ approach to cosmetic packaging is built on this field reality — not on marketing claims about technology leadership.
Of the 31 cosmetic and personal care line projects I handled in Southeast Asia and Latin America between 2018 and 2023, 14 involved brands replacing or supplementing European equipment with Chinese Tier-1 machines — the primary driver in 11 of those 14 cases was spare-parts lead time and after-sales response time, not machine performance. The performance was acceptable. The support geography was not.
— Forester Xiang, HIJ Machinery, Wenzhou, China
Frequently Asked Questions: European vs. Chinese Cosmetic Packaging Machines
❓ Are Chinese cosmetic packaging machines as good as European ones?
Chinese Tier-1 cosmetic packaging machines now match European mid-range equipment on fill accuracy (±0.5%), throughput (up to 400 tubes/min), and GMP-compliant design — the measurable gap lies in documentation maturity and Annex 11 integration for EU computerized system validation. For most emerging-market applications running below 450 units/min, the performance difference does not justify a 2–3x capital cost premium. The critical variable is supplier tier: Tier-1 Chinese manufacturers with ISO 9001:2015 certification and full IQ/OQ/PQ deliverables are fundamentally different from budget suppliers with no validation infrastructure.
❓ Do Chinese cosmetic packaging machines comply with ISO 22716?
Yes. ISO 22716 does not specify equipment origin — it requires that cosmetic manufacturing equipment be suitable for its intended use, constructed of materials that do not contaminate products, and maintained with documented procedures. A Chinese cosmetic packaging machine built with 316L stainless steel product-contact surfaces, validated cleaning procedures, and a documented preventive maintenance program fully satisfies ISO 22716 Section 4 requirements. Compliance is achieved through equipment design and qualification documentation, not country of manufacture.
❓ What is the price difference between European and Chinese cosmetic packaging machines?
European cosmetic tube filling machines in the 150–300 tubes/min range typically cost $180,000–$320,000. Equivalent Chinese Tier-1 machines run $75,000–$130,000 — a 40–55% saving on capital cost. Over a five-year total cost of ownership including spare parts, commissioning, and downtime, the gap widens further for emerging-market operators: European equipment in Southeast Asia or Latin America typically runs 2.1–2.8x higher TCO than Chinese Tier-1 equipment, driven primarily by spare-parts lead times and service infrastructure costs. Budget Chinese machines below $55,000 frequently eliminate the capital saving through higher downtime and rework costs.
❓ How long do spare parts take for European cosmetic packaging machines in Asia or Latin America?
European OEM spare parts shipped to Southeast Asia, Latin America, or Africa typically take 8–16 weeks for non-stocked components, even with air freight. Custom-engineered parts — forming dies, sealing jaws, specialized tooling — can take 12–20 weeks. Chinese Tier-1 suppliers with regional spare-parts hubs in Singapore, Dubai, or Mexico City typically fulfill the same categories in 1–4 weeks. For high-volume cosmetic lines where each day of downtime costs $6,000–$12,000, this lead-time difference is frequently the single most important procurement variable.
❓ Can a Chinese cosmetic packaging machine pass an FDA or EU GMP audit?
Yes, provided the machine was built and qualified correctly. FDA 21 CFR Part 211 and EU GMP do not specify equipment origin. A Chinese cosmetic filling machine that has been installed with documented IQ/OQ/PQ qualification, operates within validated parameters, and is maintained per a documented preventive maintenance schedule meets the equipment requirements of both regulatory frameworks. I have personally overseen six line qualifications where Chinese equipment passed FDA-guided pre-approval inspections without conditions. The documentation package — not the machine’s country of origin — determines audit outcomes.
❓ What should I ask a Chinese cosmetic packaging machine manufacturer before buying?
Six questions cut through the noise: (1) Can you provide a sample IQ/OQ/PQ package from a comparable installation? (2) What PLC platform do you use, and will full source code be transferred at FAT? (3) What is your CE marking scope — can you provide the full technical file? (4) Where is your nearest spare-parts stock for my region, and what are the typical lead times for your five highest-wear components? (5) Can you provide two references from customers in my region who have run this machine model for more than 18 months? (6) What is your FAT protocol — can I see a sample run against a customer URS? Any supplier who cannot answer all six clearly should be removed from your evaluation. See also common cosmetic packaging procurement mistakes for additional due diligence guidance.
❓ Is a European cosmetic packaging machine worth the extra cost?
In three specific scenarios: yes. (1) When throughput requirements exceed 450 units/min with sub-±0.3% fill tolerance — European platforms like IMA and Norden hold a real mechanical and software lead here. (2) When EU Annex 11 / GAMP 5 computerized system validation is mandatory and your quality team lacks the resources to close the documentation gap on a Chinese platform. (3) When your product spans pharma-cosmetic regulatory overlap and a unified FDA drug site qualification history is commercially critical. Outside these three scenarios, the additional capital cost rarely generates proportional performance or compliance returns — particularly for manufacturers operating in emerging markets where the service infrastructure for European OEMs is systematically thin.
🔗 Related Resources in This Cluster
→ Full cost breakdown: Cosmetic Packaging Machine Cost Guide — capital cost, TCO, and hidden variables across machine categories.
→ Avoid procurement traps: 7 Costly Cosmetic Packaging Mistakes — field-documented failures and how to prevent them.
→ HIJ Cosmetic Packaging Machines: Full product range — tube fillers, sachet machines, wet wipes lines, and complete turnkey solutions.
→ Turnkey cosmetic packaging lines: Integration, validation, and commissioning — how HIJ manages the full project lifecycle.
🎯 Ready to Compare Real Specifications?
Get an Honest Machine Comparison for Your Specific Line
Tell us your throughput requirements, target market, and compliance scope. I’ll tell you whether a European or Chinese machine makes more sense for your situation — and exactly what documentation to demand before you sign any PO.

