💰 Pharma Buyer’s Guide — Cost Analysis
The Real Cost of a Syringe Blister Packing Machine: What Pharma Buyers Must Know
The sticker price on a syringe blister packing machine is rarely the real cost. Total cost of ownership — including validation support, tooling changeovers, and compliance documentation — routinely doubles the initial investment for unprepared buyers. This guide breaks down every cost layer, so your procurement team knows exactly what to budget before the purchase order is signed.
⚡ Direct Answer
A syringe blister packing machine carries a quoted price of $35,000–$180,000+ depending on output speed, forming system, and GMP specification level. Total cost of ownership — when IQ/OQ/PQ validation, custom tooling, FAT/SAT travel, spare parts inventory, and first-year consumables are included — typically reaches 1.8× to 2.4× the machine price. Buyers who budget only for the machine price face line stoppages, failed inspections, and unplanned capital calls within 18 months.
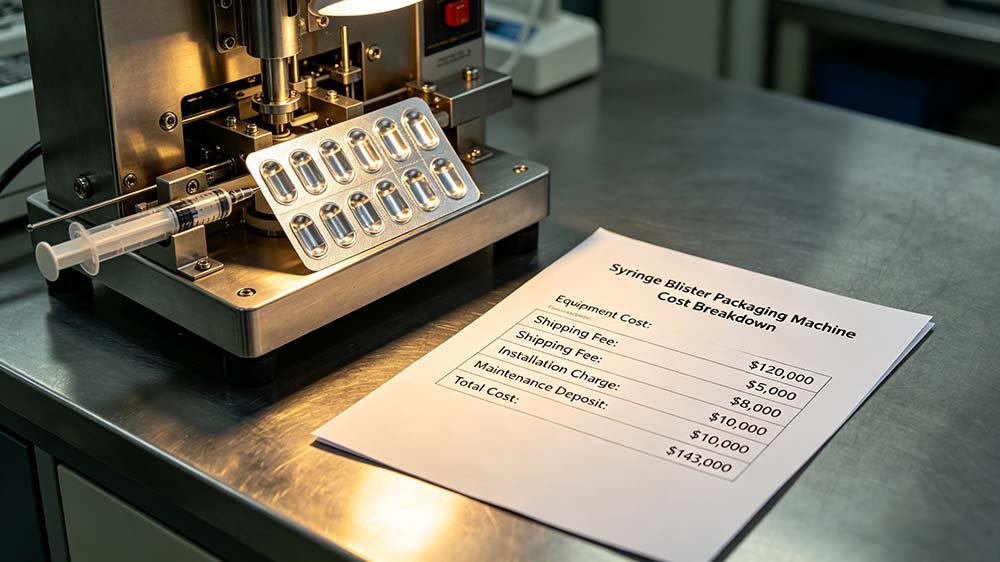
$35K–$180K+
Quoted machine price range
1.8×–2.4×
Typical TCO multiplier
12%
Pharma recalls linked to packaging (FDA 2019–2023)
6–8 months
Typical timeline lost to incomplete validation docs

Figure 1 — The full cost landscape of a syringe blister packing machine: from quoted price to true total cost of ownership.
The Number Teams Look at First Is Exactly the Wrong Number
⚡ Direct Answer
The quoted machine price for a syringe blister packing machine covers the hardware only — it excludes validation packages, custom tooling, consumables, and FAT/SAT costs that collectively add 80%–140% to the total project spend.
Procurement teams open three quotations. They rank them by machine price. The lowest number wins internal approval. That’s the process. It’s also the process that produced the six-month line stoppage I witnessed at a Mexican injectable manufacturer in 2021 — not from machine failure, but from a failed FDA pre-approval inspection triggered by missing FAT and SAT records. The vendor had quoted $52,000 for the machine. Nobody asked what validation documentation cost extra. The answer: it wasn’t offered at all.
FDA enforcement records for 2019–2023 show packaging as the root cause in 12% of pharmaceutical recalls — the category with the strongest correlation to emerging market distribution failures. That number is the one I use when a procurement director tells me the $8,000 cheaper machine justifies the trade-off. It usually ends the conversation.
The machine price tells you what the vendor wants to be compared on. Total cost of ownership tells you what the project will actually cost. They are different numbers. Often very different.
The total cost of ownership for a syringe blister packing machine runs 1.8× to 2.4× the quoted price when validation, tooling, and compliance documentation are properly accounted for — a gap that surfaces during budget review, not during quotation.
— Forester Xiang, HIJ Machinery
What Does a Syringe Blister Packing Machine Actually Cost? A Tier-by-Tier Breakdown
⚡ Direct Answer
Syringe blister packing machine prices range from $35,000 for entry-level semi-automatic configurations to $180,000+ for high-speed fully automatic lines with integrated vision inspection and cGMP servo control systems — with the most common pharma-grade mid-range units priced between $65,000 and $120,000.
Machine Price Tiers by Configuration
| Configuration Tier | Typical Quoted Price (USD) | 출력 속도 | GMP Level | Validation Package Included? |
|---|---|---|---|---|
| Entry Semi-Automatic | $35,000–$55,000 | 20–40 blisters/min | Basic GMP | No — separate cost |
| Mid-Range Automatic | $65,000–$100,000 | 60–120 blisters/min | WHO GMP / cGMP | Partial (IQ template only) |
| Full-Spec Pharma Automatic | $100,000–$145,000 | 120–180 blisters/min | WHO GMP + 21 CFR Part 11 | IQ/OQ/PQ full package |
| High-Speed Integrated Line | $145,000–$180,000+ | 180–250 blisters/min | EU GMP Annex 1 + 21 CFR | Full DQ/IQ/OQ/PQ + SAT |
| European-Brand Equivalent | $280,000–$500,000+ | 150–300 blisters/min | EU GMP Annex 1 | Full package (built into price) |
Source: HIJ Machinery project database, 2022–2025. European-brand pricing from customer-provided competitive quotations received during pre-sales consultations.
The mid-range automatic tier — $65,000–$100,000 — is where most emerging-market pharma buyers land. It covers the output speed they need (60–120 blisters/min handles most syringe SKUs at 10–25 million units/year annual volume), and the GMP specification level that passes WHO and national regulatory review. That’s the price on the quotation. Then the real conversation starts.

Figure 2 — TCO breakdown: the machine price is only the starting point for a complete syringe blister packaging investment.
The Eight Cost Layers Nobody Puts in the Quotation
⚡ Direct Answer
Beyond the machine price, syringe blister packing machine buyers face eight additional cost categories — including custom tooling ($8,000–$35,000 per syringe format), IQ/OQ/PQ validation ($15,000–$45,000), FAT/SAT travel costs, spare parts inventory, and operator training — that collectively add $50,000–$130,000 to the total project budget.
Of the 31 syringe and sterile device blister line projects I handled between 2018 and 2024, every single one had at least three cost categories that the buyer had not budgeted for at project initiation. The most common offenders were tooling, validation documentation, and site adaptation. Together they averaged $72,000 in unplanned spend — on top of machines quoted between $65,000 and $110,000.
Cost Layer 1: Custom Tooling and Forming Molds
Syringes are not tablets. The forming cavities must be engineered precisely to the syringe barrel diameter, flange geometry, needle guard profile, and plunger rod clearance. Standard tooling does not exist for syringe blister applications. Every format requires custom mold design and CNC machining. Budget $8,000–$35,000 per syringe format, depending on complexity. If your product range covers three syringe sizes, that’s $24,000–$105,000 in tooling before a single blister is produced.
Changeover tooling — the secondary tool sets needed for fast-format switching — adds another $5,000–$12,000 per additional SKU. And tooling has a lifecycle. Under normal production conditions at 80–100 blisters/min, forming molds typically require refurbishment after 8–12 million cycles. That’s a recurring cost most three-year financial models miss entirely.
Cost Layer 2: IQ/OQ/PQ Validation
This is the largest single hidden cost for buyers in regulated markets. Full IQ/OQ/PQ documentation for a syringe blister packing machine — including protocol writing, execution, deviation management, and final report compilation — runs $15,000–$45,000 depending on market requirements. FDA-regulated lines in the US or Latin American export markets tend toward the upper end. WHO GMP markets can often be served at $15,000–$25,000.
The CDSCO reviewer I dealt with on an Indian export-market project in 2022 cited ICH Q7 directly and asked why process qualification data was absent. The dossier went back. Eight months later. The validation cost was $18,000. The eight-month delay in market entry cost the client approximately $340,000 in deferred revenue by their own estimate. The ratio is instructive.
ℹ️ Regulatory Compliance Reference
GMP validation requirements for syringe blister packaging lines are governed by WHO TRS 992 부속서 3 (global), 21 CFR 파트 211 (US and US-export markets), EU GMP Annex 1 (2022 revision, sterile manufacturing), and ICH Q7 (API/device manufacturer alignment). Validation documentation must cover DQ, IQ, OQ, and PQ phases. Lines destined for FDA Pre-Approval Inspection (PAI) require FAT and SAT records as supporting evidence — these are not optional if the product is under NDA or ANDA review.
Cost Layer 3: FAT, SAT, and Travel
Factory Acceptance Testing (FAT) is conducted at the machine manufacturer’s facility before shipment. Site Acceptance Testing (SAT) happens at the buyer’s plant after installation. Both require the buyer to send qualified personnel — typically a validation engineer and a QA representative — to attend and sign off. For a buyer in Lagos or Manila traveling to Wenzhou for FAT, budget $4,000–$8,000 per person per trip. Two people, two trips. That’s $16,000–$32,000 in travel and accommodation before the machine ships.
Some vendors offer remote FAT via video link. I’m skeptical of it for syringe blister machines — the forming quality, sealing integrity, and cavity alignment checks require hands-on verification. That said, for buyers with tight travel budgets, a hybrid approach — remote FAT with a third-party witness on-site — can reduce cost by 40–60% without sacrificing documentation integrity.
Cost Layer 4: Spare Parts Inventory
WHO GMP guidelines and ICH Q10 quality system requirements both recommend maintaining a critical spare parts inventory sufficient to prevent production stoppages lasting more than 72 hours. For a syringe blister packing machine, that inventory — typically covering heating elements, sealing plates, servo drive components, forming station wear parts, and pneumatic actuators — costs $8,000–$22,000 at initial setup. This is not a one-time cost; spare parts replenishment runs $3,000–$6,000 annually for a line running two shifts.
Cost Trap Diagnostics: Three Scenarios That Blow the Budget
🔴 Symptom: Validation cost not in vendor scope
Root cause: Vendor quoted hardware only. IQ/OQ/PQ protocols, execution, and reporting are treated as professional services sold separately — sometimes by a third-party CRO that charges $200–$350/hour.
→ Fix: Require a line-item validation scope in the commercial proposal. If the vendor cannot provide it, request a written statement of what documentation they will deliver and obtain third-party CRO quotes before signing.
🔴 Symptom: PLC protocol incompatible with existing MES
Root cause: Machine uses a proprietary or non-standard PLC architecture that cannot communicate with the buyer’s Manufacturing Execution System. 21 CFR Part 11 electronic records requirements may also be unmet if audit trail functionality is absent.
→ Fix: Specify PLC brand (Siemens, Allen-Bradley, or equivalent) and MES interface protocol (OPC-UA preferred) in the URS before quotation. Confirm audit trail functionality explicitly.
🔴 Symptom: Tooling cost quoted as “TBD”
A vendor who cannot provide tooling pricing before contract signature has not yet designed the mold. That means your format-specific syringe geometry — including needle guard and flange — has not been reviewed. Tooling underquotes of $15,000–$40,000 are common when this step is deferred.
→ Fix: Provide syringe dimensional drawings (barrel OD, flange OD, overall length, needle guard profile) at RFQ stage. Require firm tooling pricing before signing.
Cost Layers 5–8: Installation, Training, Consumables, and Regulatory Fees
Installation and commissioning — sending 1–2 engineers to the buyer’s site for 5–10 days — runs $8,000–$18,000 including travel when the buyer is in Southeast Asia, Africa, or Latin America. Operator training for 3–5 production staff typically takes 3–5 days and costs $3,000–$8,000 as a separately invoiced service. First-year consumables — forming film, lidding foil, lubricants, cleaning agents — add $12,000–$28,000 depending on run volumes. Finally, regulatory submission fees and testing lab costs for packaging validation studies (container closure integrity per USP <1207>, moisture vapor transmission) can add $5,000–$15,000 for markets with strict pre-market approval requirements.
The Conversation That Changed How I Explain This
📋 Forester’s Insight — A 20-Year Engineer’s Perspective
A procurement director in São Paulo called me in 2023. She had three quotations in front of her: $58,000, $74,000, and $88,000. Her finance team had already pre-approved the $58,000 option. She called because something felt off. I asked her to read me what the $58,000 quotation included. Machine, forming mold (standard), electrical installation diagram, and one-year warranty on parts. That was the complete scope. I asked what it excluded. She went quiet for a moment. Then: “It doesn’t mention validation at all.”
We spent 40 minutes on that call. By the end, her all-in budget for the $58,000 machine — after adding validation, custom tooling for her 1mL and 5mL syringe formats, FAT travel, spare parts, and installation — was $127,000. The $88,000 machine with a full IQ/OQ/PQ package and custom tooling included landed at $119,000 total. She chose the $88,000 machine. I’m still not entirely sure I convinced her — I think the math convinced her.
The strategic point: before signing anything, demand a full URS review and confirm cGMP compliance documentation is contractually guaranteed. Our turnkey integration approach means validation packages, tooling support, and cross-system compatibility are built into scope from day one — not invoiced as afterthoughts six months into the project.

Figure 3 — Forester Xiang with the HIJ engineering team during a pre-FAT cost review session for a Southeast Asian syringe packaging project.
Syringe Blister Packing Machine Total Cost of Ownership: Three Supplier Profiles Compared
⚡ Direct Answer
Across three common supplier profiles — standard Chinese exporter, GMP-spec Chinese manufacturer like HIJ, and European brand — the total 3-year cost of ownership ranges from $98,000 to $620,000 for a comparable syringe blister packaging line, with the primary differentiators being validation depth, tooling quality, and post-sale service infrastructure.
| Cost Category | Standard Chinese Exporter | HIJ Machinery (GMP-Spec) | European Brand |
|---|---|---|---|
| Machine Price | $35,000–$58,000 | $65,000–$120,000 | $280,000–$480,000 |
| Custom Tooling (2 formats) | $18,000–$30,000 | $16,000–$28,000 | $28,000–$55,000 |
| IQ/OQ/PQ Validation | $25,000–$45,000 (3rd party) | $0 (included in scope) | $0 (included in price) |
| FAT/SAT & Installation | $18,000–$28,000 | $10,000–$16,000 | $22,000–$38,000 |
| Spare Parts (Year 1) | $10,000–$18,000 | $8,000–$14,000 | $18,000–$35,000 |
| Year 2–3 Maintenance & Parts | $20,000–$35,000 | $14,000–$22,000 | $30,000–$60,000 |
| Estimated 3-Year TCO | $126,000–$214,000 | $113,000–$200,000 | $378,000–$668,000 |
| GMP Documentation Level | Basic / none | WHO GMP + 21 CFR Part 11 | EU GMP Annex 1 + 21 CFR |
| Validation Package | None included | Full DQ/IQ/OQ/PQ | Full DQ/IQ/OQ/PQ |
| On-Site Service Response | 30–90 days (parts-dependent) | 14–21 days (global network) | 7–14 days (Europe-centered) |
Note: TCO figures represent estimated ranges based on HIJ project data and customer-disclosed competitive quotations from 2022–2025. Actual costs vary by syringe format complexity, target regulatory market, and buyer location. European brand pricing derived from customer-shared RFQ responses; not from official published pricing.
The table makes one point clearly. A standard Chinese exporter with a $45,000 machine price does not deliver the lowest TCO — because the $25,000–$45,000 third-party validation cost and the higher spare parts failure rate close the gap fast. That’s before accounting for the six-to-twelve-month validation delay that occurs when the vendor cannot support your QA team through IQ/OQ execution. At $340,000 in deferred revenue per eight months (the São Paulo project estimate I referenced earlier), the validation delay alone dwarfs the hardware savings.
How to Build a Realistic Syringe Blister Packaging Budget Before You Issue an RFQ
⚡ Direct Answer
A realistic syringe blister packing machine budget requires five line items beyond the machine price: tooling per format, validation package (or third-party validation budget), FAT/SAT and installation, first-year spare parts, and consumables — adding a minimum 60%–90% to the machine price for a properly scoped project.
The budget report landed in March. Two line items: machine ($78,000) and installation ($5,000). The project lead had never built a pharma packaging line before. He had no reason to know what was missing. He got the approval. Six months later, he was back in front of the finance committee asking for another $68,000 to cover validation, tooling for the second syringe format, and the replacement servo drive that failed at month four. That meeting was not comfortable.
Here is a structured budget planning checklist for any pharma buyer approaching a syringe blister machine purchase:
Machine price (base): Confirm this is an FOB Wenzhou or CIF your-port price. Shipping, customs, and local import duties add 8–15% in most emerging markets.
Custom tooling per syringe format: Request firm pricing with mold drawings. Budget $8,000–$35,000 per format. If you have 3+ syringe sizes, this line item can exceed the machine price.
IQ/OQ/PQ validation package: Confirm whether it is included in vendor scope or priced separately. If separate, budget $15,000–$45,000 depending on regulatory market and documentation depth required.
FAT travel costs: Budget $4,000–$8,000 per person per trip. Two people for FAT minimum. Factor visa processing time (4–8 weeks for some markets).
SAT and commissioning: Vendor engineer on-site 5–10 days. Budget $8,000–$18,000 including travel from China.
Spare parts initial inventory: At minimum: forming mold wear inserts, sealing plate set, key pneumatic components, heating elements. Budget $8,000–$22,000.
First-year consumables: Forming film (PVC/Tyvek/Alu-Alu depending on format), lidding foil, lubricants. Budget based on planned production volume — typically $12,000–$28,000/year at medium throughput.
Operator training: 3–5 staff, 3–5 days. Budget $3,000–$8,000 if vendor charges separately. Confirm language support (English, Arabic, Spanish, Portuguese, Bahasa).
Regulatory testing and submission fees: Container closure integrity testing (ASTM F2338 / USP <1207>), MVTR testing, and any pre-market approval submission costs for your target market. Budget $5,000–$15,000.
Contingency (15–20% of total): Format qualification failures, additional stability testing, re-validation after site modification. This is not optional padding — it is statistically necessary based on project completion data.
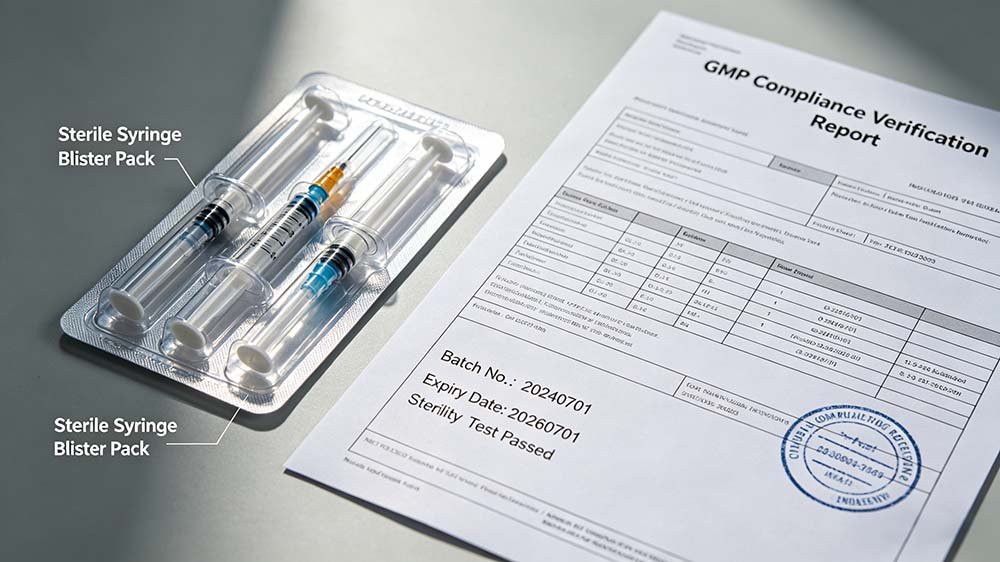
Figure 4 — A fully GMP-compliant syringe blister packaging line mid-IQ/OQ qualification — the validation phase most cost budgets underestimate or omit entirely.
How Syringe Blister Machine Costs Vary by Market: Southeast Asia, Middle East, Africa, and Latin America
⚡ Direct Answer
Market-specific factors — import duties (8–30% depending on country), local regulatory submission costs, infrastructure adaptation requirements, and regional service response time — add $15,000–$55,000 in market-specific costs on top of the base TCO for buyers in emerging pharma markets.
Three weeks into a job in Jakarta in 2019, I got a call from the client’s engineering manager. The local electrical standard required a panel rewire — Indonesian PLN grid voltage and frequency tolerance demanded an isolation transformer the quotation hadn’t included. $4,200. Not a large number. But it hadn’t been budgeted, it delayed commissioning by nine days, and it was the third unbudgeted item that week. The project team’s confidence was already fragile.
Regional cost variables to build into your budget:
| Market Region | Import Duty (Machinery) | Local Regulatory Costs | Infrastructure Adaptation | Service Response (HIJ) |
|---|---|---|---|---|
| Southeast Asia (Indonesia, Vietnam, Thailand) | 5–15% | $5,000–$18,000 | Voltage adaptation common; IP rating for humidity | 10–18 days |
| Middle East (Saudi Arabia, UAE, Egypt) | 5–12% | $8,000–$22,000 | Dust-rated panels; cooling in high-temp environments | 14–21 days |
| Sub-Saharan Africa (Nigeria, Kenya, South Africa) | 10–30% | $10,000–$28,000 | Voltage stabilization; UPS requirements common | 18–28 days |
| Latin America (Brazil, Mexico, Colombia) | 12–25% | $12,000–$35,000 | ANVISA/COFEPRIS documentation; Spanish/Portuguese docs | 14–21 days |
Duty rates are approximate and subject to HS code classification and bilateral trade agreements. Regulatory costs reflect typical national agency submission and testing fees as of 2024–2025. Confirm current rates with a local customs broker and regulatory affairs consultant before final budget approval.
Brazil’s ANVISA and Mexico’s COFEPRIS both require locally-submitted GMP documentation for imported packaging equipment used in sterile drug manufacturing. For a syringe blister line, that means your validation package must be translated, reviewed, and sometimes re-executed with ANVISA-witnessed protocols. Budget an additional $12,000–$28,000 for Latin American regulatory submission costs beyond the standard IQ/OQ/PQ package. For more context on how these regional challenges affect total project planning, see our guide on syringe blister packaging GMP compliance and validation requirements.
How to Justify the Investment: ROI Framing for Syringe Blister Packaging Lines
⚡ Direct Answer
A syringe blister packing machine with a 3-year TCO of $130,000–$200,000 typically reaches ROI breakeven in 14–22 months for pharma manufacturers running 8–15 million syringes/year — driven by labor savings (4–6 operators replaced per shift), rework reduction (from 3–6% manual pack reject rate to 0.1–0.5% automated), and regulatory compliance protection against recall risk.
Of the 31 syringe packaging projects I handled in Southeast Asia and Africa between 2018 and 2024, the clients who documented ROI consistently cited three drivers: labor cost savings averaging $85,000–$140,000 annually (replacing 4–6 manual packaging operators at two-shift production), rework cost elimination (manual packing reject rates of 3–6% drop to 0.1–0.5% with automated cavity sensing and vision inspection), and regulatory risk reduction. That last driver is harder to quantify but is not zero.
A batch recall of a sterile injectable product costs $800,000–$4,000,000 in direct costs (product write-off, logistics, regulatory response, brand impact) per FDA and industry estimates. If poor packaging integrity is the root cause — and it is the root cause in 12% of pharmaceutical recalls per FDA enforcement data — then the $130,000 machine that prevents a single recall event pays for itself in a fraction of its operational life. I won’t tell you the math is simple. It involves your API profit margins, your volume, your target markets, and your regulatory exposure. But I’ve never seen a client who properly modeled the recall risk scenario and then chose the cheapest machine.
A properly specified syringe blister packing machine, with full validation and custom tooling, pays back its total cost of ownership in 14–22 months through labor savings and rework elimination alone — before accounting for regulatory compliance protection worth multiples of the hardware investment.
— Forester Xiang, HIJ Machinery
Red Flags in Quotations: What to Reject Before Signing
🔴 Validation scope is blank or “as required by client”
This language shifts all validation responsibility to you. The vendor will provide no protocols, no execution support, and no deviation management. You will need a CRO at $200–$350/hour.
→ Reject: Require a specific validation scope statement listing which documents the vendor prepares, executes, and signs.
🔴 Tooling quoted as “standard” for syringe application
No standard tooling exists for syringe blister packaging. A vendor quoting “standard” forming molds has not reviewed your syringe geometry. The mold will need modification post-delivery — at your cost and on your timeline.
→ Reject: Require mold engineering drawings reviewed against your syringe dimensional spec before contract signature.
For a deeper look at what a complete turnkey syringe packaging project includes — from URS through FAT to production qualification — see our detailed breakdown: what’s included in a turnkey syringe blister packaging line. Understanding the full scope is the most reliable way to avoid the cost surprises described in this article.
Frequently Asked Questions: Syringe Blister Packing Machine Cost
How much does a syringe blister packing machine cost?
A syringe blister packing machine costs $35,000–$180,000+ depending on output speed, automation level, and GMP specification. The most common pharma-grade automatic configuration for emerging markets runs $65,000–$120,000 for the machine alone. Total cost of ownership including tooling, validation, installation, spare parts, and consumables typically reaches $113,000–$200,000 over the first three years of operation.
What is included in the validation cost for a syringe blister packaging machine?
Full IQ/OQ/PQ validation covers four phases: Design Qualification (DQ) confirming the machine meets URS requirements; Installation Qualification (IQ) verifying correct installation; Operational Qualification (OQ) demonstrating the machine performs within specification across its operational range; and Performance Qualification (PQ) confirming consistent output under production conditions. Protocol writing, execution, deviation management, and final report preparation are included. Third-party validation costs $15,000–$45,000. HIJ Machinery includes full DQ/IQ/OQ/PQ in scope for GMP-spec line orders.
Why is custom tooling so expensive for syringe blister packs?
Syringe blister forming cavities must accommodate syringe barrel diameter, flange geometry, plunger rod clearance, and needle guard profile — all format-specific variables that require CNC-machined precision tooling. No universal standard exists. Each syringe format requires a dedicated mold design review, engineering, and machining cycle of 4–8 weeks. At $8,000–$35,000 per format, tooling costs for a 3-SKU syringe range can exceed the machine price.
Can I get a cheaper machine and hire a CRO for validation separately?
Yes. But the math rarely favors it. A CRO validation engagement for a syringe blister line costs $25,000–$45,000 at $200–$350/hour rates, and takes 3–6 months because the CRO must first understand the machine — work the vendor would have done internally. Total TCO for the cheaper machine plus CRO validation typically equals or exceeds a GMP-spec machine with validation included, while adding significant timeline risk. One project delay of 6 months on a product with $1M/year revenue potential costs $500,000 in deferred income.
What is the ROI timeline for a syringe blister packing machine?
Breakeven typically occurs in 14–22 months for plants running 8–15 million syringes/year on a two-shift schedule. Primary ROI drivers: labor savings ($85,000–$140,000/year replacing 4–6 manual operators), rework elimination (reject rate from 3–6% to 0.1–0.5%), and reduced recall risk. Plants with higher volumes or targeting export markets with strict GMP requirements see faster payback because compliance protection preserves market access worth far more than the machine investment.
Does import duty significantly affect total machine cost?
Yes — especially in Africa and Latin America. Import duties on pharmaceutical packaging machinery range from 5% (ASEAN preferential rates) to 30% (some Sub-Saharan African markets). On a $90,000 machine, a 25% duty adds $22,500 before the machine reaches your facility. Add freight, insurance, and local handling, and landed cost can be 35–40% above FOB price. Always request CIF pricing and confirm HS code classification with your customs broker before finalizing the budget.
How do I know if a quotation is missing hidden costs?
Check for these five items in the quotation scope. If any are absent or marked “TBD,” treat the quotation as incomplete: (1) custom tooling pricing with format-specific mold drawings; (2) validation scope statement specifying which DQ/IQ/OQ/PQ documents the vendor delivers; (3) FAT and SAT terms including who bears travel costs; (4) spare parts list with initial inventory recommendation; (5) software license terms for any 21 CFR Part 11–compliant audit trail or SCADA system. A quotation missing any of these five will deliver budget surprises.
🎯 Ready to Build Your Real Budget?
Get a Complete Cost Breakdown — Machine + Tooling + Validation Included
Stop guessing at hidden costs. Our engineering team will review your syringe format specifications, target regulatory market, and production volume — then provide a full-scope quotation covering machine, custom tooling, IQ/OQ/PQ validation, FAT/SAT, and first-year support. No line items marked “TBD.”