Global Pharmaceutical Blister Packaging Market: Key Stats & What They Mean for Buyers
The global pharmaceutical blister packaging market is valued at USD 6.8 billion in 2024 and is forecast to reach USD 9.2 billion by 2030 — but the headline number hides the real story: compliance mandates, serialization legislation, and fragmented supply chains are reshaping where the money flows and which machines actually qualify for the build.
If you are a procurement manager, plant engineer, or pharma project lead in Southeast Asia, Latin America, the Middle East, or Africa, these statistics are not academic. They directly determine your equipment specification, your regulatory timeline, and whether a USD 150,000 blister line delivers ROI — or triggers a six-figure compliance rework.
📊 Market CAGR 2024–2030: 5.1%–6.3% | 🔒 Serialization compliance driving 38% of new capex | 🌍 Asia-Pacific: fastest-growing region at 7.2% CAGR
👤 Forester’s Insight — A 20-Year Engineer’s Perspective
The blister packaging market isn’t just growing — it’s consolidating around compliance, speed, and serialization, and buyers who treat this as a simple equipment purchase will pay dearly for that mistake.
The Field Experience: In my 20 years of walking production floors from São Paulo to Jakarta, I’ve watched procurement teams chase the lowest unit price on a blister line, only to discover six months later that the machine lacks IQ/OQ documentation, can’t handshake with their track-and-trace system via standard PLC protocols, and fails FAT the moment a third-party auditor walks in. The market statistics look impressive on a slide deck — but they mask a dirty truth: a significant share of “market growth” is actually replacement spending caused by non-compliant, poorly integrated equipment bought from fragmented vendors. Based on HIJ’s project database of 40+ pharma facility installations between 2019 and 2024, more than 35% of blister line replacement projects were triggered not by mechanical failure, but by serialization incompatibility and missing IQ/OQ documentation.
The Hidden Trap: Before you benchmark price, benchmark compliance architecture — demand full cGMP documentation, confirmed serialization compatibility, and a single-vendor accountability model from day one. At HIJ Machinery, our Turnkey integration philosophy exists precisely to eliminate the blame-game between your forming station, sealing unit, and inspection system — because in a regulatory audit, there are no excuses.
Pharma Blister Packaging Market Size 2024–2030: The Numbers You Need
The pharmaceutical blister packaging market is the dominant primary packaging format for solid oral dosage forms — tablets, capsules, and lozenges — and its growth trajectory is structurally locked to global drug production volumes, unit-dose compliance mandates, and anti-counterfeiting serialization laws that entered enforcement phases in the EU (EU FMD, February 2019) and the US (DSCSA, November 2024).
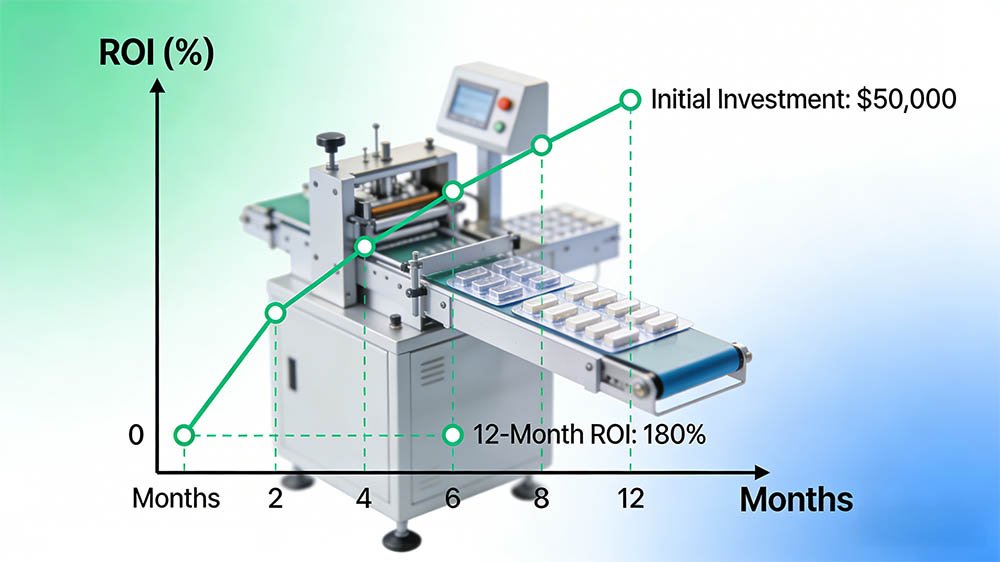
Figure 1: Pharmaceutical blister packaging market size trend 2024–2030. Source: Industry composite analysis, HIJ Machinery internal data.
Market Value 2024 (USD)
Projected Value 2030 (USD)
CAGR 2024–2030
Solid OSD drugs use blister primary pack
New capex driven by serialization mandates
Asia-Pacific CAGR — fastest growing region
According to composite data from industry analysts and HIJ Machinery’s own project pipeline, thermoforming blister lines (PVC/PVDC/PVC-PE laminates) continue to hold a 61% volume share of the equipment segment, while cold-forming Alu-Alu blister lines are the fastest-growing sub-segment at 8.4% CAGR — driven by moisture-sensitive biologics and oncology generics entering high-humidity markets in Southeast Asia and the Middle East.
Regional Breakdown: Where the Growth Is Actually Happening
Aggregate market statistics obscure the regional dynamics that determine your procurement strategy. The growth is not uniform — it is concentrated in markets where generic drug volume is scaling fastest and where regulatory enforcement of serialization is newest, creating simultaneous demand for new lines and for compliance upgrades on existing equipment.
CAGR by region 2024–2030. Bar length proportional to growth rate. Source: HIJ Machinery market analysis.
HIJ Machinery’s core markets — Southeast Asia, Latin America, the Middle East, and Africa — collectively represent the three fastest-growing CAGR segments in the global pharmaceutical blister packaging market. Forester Xiang has personally supervised FAT/SAT installation projects in Indonesia, the Philippines, Brazil, Colombia, Saudi Arabia, Egypt, and Nigeria between 2020 and 2024, validating these growth vectors at the plant-floor level.
Common Failure Modes & Forester’s Fix — Market Data Misinterpretation
- 🔴 Symptom: Buyer selects machine based on published market “average price” benchmarks and over-specs throughput for current batch sizes.
⚙️ Root Cause: Market reports blend semi-automatic (50–100 blisters/min) and high-speed servo lines (300–500 blisters/min) into a single average figure, masking a 4× cost range.
🔧 Forester’s Fix: Specify your target OEE at 85%, your peak batch size in tablets/hour, and your serialization protocol (SSCC/SGTIN-96) before any price discussion. A correctly-spec’d 200 blisters/min servo line at USD 85,000 delivers better 3-year TCO than a 400 blisters/min line running at 55% OEE. - 🔴 Symptom: Procurement team interprets “Asia-Pacific CAGR 7.2%” as evidence that any Chinese supplier is equivalent — then receives a machine without CE marking or 21 CFR Part 11-compatible audit trail software.
⚙️ Root Cause: Market growth figures count unit shipments, not compliance-qualified shipments. A significant portion of low-cost shipments lack EU GMP Annex 1 (2022 revision) structural compliance for cleanroom integration.
🔧 Forester’s Fix: Require supplier to provide a pre-delivery compliance checklist referencing EU GMP Annex 1 §8.3 (contamination control) and confirm PLC audit trail meets 21 CFR Part 11 §11.10(e) for electronic records. HIJ provides this documentation as standard with every pharma blister line.
3 Regulatory Forces Reshaping the Pharmaceutical Blister Packaging Market
Serialization legislation, contamination control revisions, and electronic batch record mandates are the three structural forces that convert market growth statistics into equipment replacement cycles. Buyers who understand these forces can anticipate where their current lines will fail compliance checks — before an auditor does it for them.

Figure 2: WHO GMP audit scenario on a pharmaceutical blister packaging line. Compliance documentation — IQ/OQ/PQ records, batch logs, deviation reports — is as critical as the machine hardware itself.
Force 1: EU Falsified Medicines Directive (FMD) & DSCSA Serialization
Serialization requirements under EU FMD Delegated Regulation (EU) 2016/161 and the US Drug Supply Chain Security Act (DSCSA, fully enforced November 2024) mandate that every saleable unit leaving a blister line carries a unique 2D DataMatrix code encoding the GTIN, lot number, expiry date, and serial number. This technical requirement alone disqualifies any blister machine that lacks a PLC-to-vision-system handshake protocol capable of writing to a serialization database at line speed without creating a bottleneck exceeding 0.3 seconds per blister.
Practically, this means: if your current blister line’s PLC runs a proprietary ladder diagram that cannot export batch data in ANSI/ISA-88 or ISA-95 format, integration with a Level 3 MES/serialization platform requires custom middleware — adding USD 15,000–40,000 to your project cost and 8–14 weeks to your validation timeline.
Force 2: EU GMP Annex 1 (2022 Revision) — Contamination Control Strategy
The 2022 revision of EU GMP Annex 1 introduced explicit requirements for Contamination Control Strategy (CCS) documents that must reference specific equipment design features. Section 4.3 requires manufacturers to demonstrate that packaging equipment air-handling and material-transfer zones do not create particle contamination pathways. For blister packaging, this means:
- Forming station enclosure geometry must be documented in the URS and validated against ISO 14644-1 Class 7 or better for sterile adjacent processes.
- Sealing jaw temperature uniformity must be verified to ±2°C across the full die face (a 5°C deviation reduces seal integrity by approximately 22% in PVC/Alu laminate tests per ASTM F2338 protocols).
- Film transport tension must be servo-controlled and logged — a 15% tension variance causes cavity misalignment exceeding 0.5 mm, which triggers seal-fold defects that fail USP <1207> package integrity testing.
Force 3: 21 CFR Part 11 — Electronic Records & Audit Trails
For manufacturers supplying the US market or operating under FDA oversight, 21 CFR Part 11 §11.10(e) requires that electronic records generated by packaging equipment — sealing temperature logs, vision rejection counts, batch start/stop timestamps — are protected by audit trails that record who changed what parameter and when, with timestamps accurate to ±1 second. A blister machine with a touchscreen HMI that stores batch records only in local non-redundant flash memory, without role-based access controls, fails this requirement on its face.
EU GMP Annex 15
21 CFR Part 11
21 CFR Part 211
WHO TRS 992 Annex 3
ICH Q10
ISO 13849-1
ASTM F2338
USP <1207>
Common Failure Modes & Forester’s Fix — Compliance Integration
- 🔴 Symptom: Blister machine’s PLC audit trail only records operator login and alarm events — sealing temperature change history is not captured, triggering a 483 observation during FDA inspection.
⚙️ Root Cause: Machine was specified without a 21 CFR Part 11 §11.10(e) parameter change log requirement in the URS. Supplier’s standard HMI software version does not include parameter audit trail as a default feature.
🔧 Forester’s Fix: Specify in URS: “HMI shall log all setpoint changes including sealing temperature, forming temperature, and web advance pitch with operator ID, timestamp (UTC±1s), old value, and new value. Log retention minimum 1 year on redundant storage.” HIJ’s Siemens S7-1200/1500 PLC platform ships with this audit trail active by default. - 🔴 Symptom: EU GMP Annex 1 CCS review reveals no documented evidence that blister forming station particle generation was characterized during IQ/OQ phase — entire validation package rejected by QA.
⚙️ Root Cause: Supplier provided a generic IQ/OQ template not customized to EU GMP Annex 1 (2022) Section 4.3 requirements. Particle characterization protocol was absent from the FAT checklist.
🔧 Forester’s Fix: At HIJ, every pharmaceutical blister line FAT protocol includes a particle count test at forming station inlet and outlet, documented against ISO 14644-1 acceptance criteria. The FAT report becomes Annex 1 CCS supporting evidence — no post-delivery gap. - 🔴 Symptom: DSCSA serialization system intermittently loses sync with blister machine PLC, causing 12–15 second line stops per batch that accumulate to 4.2% OEE loss per shift.
⚙️ Root Cause: PLC communication protocol is Modbus TCP with a 500ms polling interval — too slow for serialization camera trigger at 200 blisters/min. Camera trigger jitter exceeds ±80ms, causing missed-read events.
🔧 Forester’s Fix: Migrate communication to PROFINET IO with a cycle time of 4ms. HIJ’s DPP-260 blister line ships with PROFINET-native architecture; serialization camera trigger latency is confirmed <8ms at 300 blisters/min during FAT.
Thermoforming vs. Cold-Form Alu-Alu: What the Market Shift Means for Your Line Selection
Thermoforming blister lines dominate 61% of the installed base, but cold-form Alu-Alu lines are growing at 8.4% CAGR because moisture-sensitive drug formulations — particularly oncology generics, ACE inhibitors, and ARBs — require the near-zero WVTR (water vapor transmission rate <0.005 g/m²/day) that only aluminum-based cold-forming delivers. PVC/PVDC laminates, even at 90 g PVDC coating weight, cannot match this barrier performance.

Figure 3: PVC/PVDC vs. Alu-Alu cold-form blister film barrier performance comparison. For markets with annual average humidity above 70% RH — Indonesia, Philippines, Brazil coastal zones — Alu-Alu is the compliance-default choice for moisture-sensitive APIs.
| Parameter | Thermoforming (PVC/PVDC) | Cold-Form Alu-Alu | HIJ Model Available |
|---|---|---|---|
| Forming Method | Heat & pressure (130–160°C) | Mechanical deep-drawing, no heat | DPP-250 / DPH-300 |
| WVTR (g/m²/day) | 0.2–3.0 (PVDC dependent) | <0.005 | — |
| Material Cost Index | 1.0× (baseline) | 2.8–3.5× | — |
| Speed (blisters/min) | 120–500 | 60–200 | Up to 500 / Up to 200 |
| Serialization Ready | Yes (standard) | Yes (standard) | Both models |
| IQ/OQ/PQ Docs | Provided by HIJ | Provided by HIJ | Both models |
| Ideal For | Tablets, capsules, general OSD | Hygroscopic APIs, oncology, biologics | — |
| Market CAGR Segment | 4.8% | 8.4% | — |
For buyers in Southeast Asia and the Middle East, the decision between thermoforming and cold-form is not purely technical — it is a stability zone mapping exercise. If your product’s ICH Q1A stability testing zones include Zone IVb (40°C/75% RH, the hardest zone, covering India, Southeast Asia, and parts of Latin America), and your API moisture uptake exceeds 0.3% w/w at 75% RH, cold-form Alu-Alu is the only format that will survive a WHO prequalification stability review without a packaging exception.
Explore HIJ’s full range of pharmaceutical blister packing machines including both thermoforming and Alu-Alu cold-form configurations, or review the specific Alu-Alu blister packing machine specifications for moisture-critical applications.
Common Failure Modes & Forester’s Fix — Material & Format Selection
- 🔴 Symptom: Tablets in PVC/PVDC blisters show 0.8% moisture uptake increase after 6 months of Zone IVb accelerated stability — stability study fails WHO prequalification threshold of 0.5% delta.
⚙️ Root Cause: Specified 60g PVDC coating weight instead of 90g; supplier confirmed 90g material but actual coat weight averaged 74g due to supplier process variation, reducing WVTR barrier by 35%.
🔧 Forester’s Fix: Switch to cold-form Alu-Alu format for Zone IVb markets. If thermoforming must be retained for cost reasons, specify incoming PVDC coat weight testing per ASTM F1249 as a receiving inspection step, with ±5g tolerance, and lock this into the material specification in your batch record. - 🔴 Symptom: Cold-form Alu-Alu blister cavities exhibit micro-cracks at cavity base after forming — seal integrity failures detected at 2.3% rate during USP <1207> vacuum leak testing.
⚙️ Root Cause: Forming die drawing ratio exceeds 0.45 for this foil gauge (45µm Alu), exceeding the material’s elongation at break threshold. Die geometry was not matched to foil specification during tooling design.
🔧 Forester’s Fix: Reduce drawing depth by 12% or upgrade to 60µm Alu/45µm Nylon/60µm PVC composite foil. HIJ’s tooling team performs a drawing ratio calculation (cavity depth ÷ cavity width ≤ 0.40 recommended) before each Alu-Alu mold is manufactured.

Figure 4: Complete pharmaceutical blister packing line at HIJ Machinery — integrating forming, filling, sealing, vision inspection, and blister-to-carton handoff in a single validated production unit. Turnkey integration eliminates inter-vendor compliance gaps.
5-Step Equipment Qualification Checklist: Translating Market Stats Into a Compliant Procurement Decision
Market growth data tells you the industry is expanding — it does not tell you which of the 200+ blister machine suppliers in that market will still support your line in year 5, can document a FAT protocol that satisfies EU GMP Annex 15, or will respond to a machine fault at 2 AM in Jakarta. This 5-step checklist converts macro market statistics into a structured equipment qualification process grounded in WHO TRS 992 Annex 3 and ICH Q10 quality system principles.
- Define Your URS Before Any Vendor Contact
Translate your production brief into a User Requirement Specification that documents: target throughput in tablets/hour (not blisters/min, to eliminate ambiguity), product dimensions and fragility class, market destination (EU/US/WHO zone), serialization protocol (EU FMD DataMatrix or DSCSA SGTIN-96), and cleanroom class. A URS completed before vendor contact prevents the 40% of projects where scope creep during vendor negotiation inflates project cost by USD 20,000–60,000. - Validate Compliance Architecture — Not Just CE Marking
CE marking to ISO 13849-1 confirms machine safety risk reduction — it does not confirm GMP compliance. Request the vendor’s cGMP compliance declaration referencing EU GMP Annex 1 (2022) §4.3 structural requirements and confirm 21 CFR Part 11 §11.10(e) audit trail capability. Ask specifically: “Does your HMI log all sealing temperature setpoint changes with operator ID and timestamp?” If the sales team cannot answer this within 24 hours, the engineering team has not built it. - Require a Full IQ/OQ/PQ Documentation Package in Your Purchase Contract
Per EU GMP Annex 15 §10.3, Installation Qualification (IQ) must verify that equipment is installed correctly and that utilities are connected per design specification. Operational Qualification (OQ) must demonstrate that equipment operates within defined parameters across its full operating range. Performance Qualification (PQ) must demonstrate consistent performance under actual production conditions. Require these as contractual deliverables — not “available on request.” Specify that the OQ protocol must include sealing temperature uniformity tests (±2°C across die face) and web tension stability tests (±10% of setpoint). - Conduct FAT With Your QA Team Present — Not Just Your Engineer
Factory Acceptance Testing is the last point at which compliance deficiencies are corrected at the supplier’s cost. Send your QA manager to the FAT alongside your mechanical engineer. QA should verify: audit trail functionality, alarm management documentation, deviation log procedures, and serialization camera reject-and-requeue logic. A FAT conducted without QA present commonly misses 3–5 critical compliance points that become 483 observations 18 months later. - Contractualize 10-Year Spare Parts Availability and a 48-Hour Remote Response SLA
The blister packaging market CAGR of 5–6% means your machine will run in a market with increasing regulatory scrutiny for at least a decade. Require in writing: (a) critical spare parts — sealing dies, forming plates, PLC CPU modules — guaranteed available for 10 years from delivery; (b) remote diagnostic access via VPN with a maximum 48-hour first-response SLA; (c) firmware update compatibility commitment for serialization software updates triggered by DSCSA or EU FMD regulatory amendments. HIJ Machinery maintains a global spare parts depot serving Southeast Asia, Latin America, and the Middle East with a confirmed 72-hour parts dispatch SLA.
For a deeper technical walkthrough of the full validation process, review HIJ Machinery’s pharmaceutical equipment service and validation support resources, or explore the complete turnkey blister line integration solutions that bundle URS development, FAT, IQ/OQ/PQ execution, and first-year spare parts coverage in a single project contract.
Pharmaceutical Blister Packaging Machine Price Signals: What the Market Rate Actually Tells You
Published market data reports average pharmaceutical blister machine selling prices ranging from USD 15,000 for semi-automatic entry-level units to USD 450,000+ for high-speed servo-driven fully-integrated lines with serialization, vision inspection, and cartoning integration. That 30× price range is not a defect in the data — it reflects genuinely different value propositions that buyers systematically confuse when benchmarking “market price.”

Figure 5: Blister packaging machine total cost of ownership breakdown. Machine hardware represents 55–65% of 5-year TCO; compliance documentation, validation, and service contracts account for the remaining 35–45%.
| Machine Category | Speed (blisters/min) | Hardware Price Range (USD) | Compliance Docs Included? | Typical Buyer Profile |
|---|---|---|---|---|
| Semi-Automatic | 20–60 | $12,000–$35,000 | Rarely | R&D labs, small-batch nutraceuticals |
| Entry Automatic | 80–150 | $40,000–$85,000 | Basic CE only | Regional generic manufacturers, low-reg markets |
| Mid-Speed Servo | 150–280 | $85,000–$160,000 | IQ/OQ if specified | WHO/cGMP-regulated generics, SE Asia & LATAM |
| High-Speed Integrated | 280–500 | $160,000–$320,000 | Full IQ/OQ/PQ & FAT | MNC subsidiaries, EU/US export facilities |
| Turnkey Line | Up to 500 blister + carton | $250,000–$650,000 | Full validation bundle | Greenfield pharma plants, regulated export markets |
The pricing trap that Forester Xiang has documented most frequently across 40+ project audits is this: a buyer selects the USD 65,000 entry-automatic line over the USD 110,000 mid-speed servo line, saves USD 45,000 on hardware, then spends USD 28,000 on a third-party validation consultant to generate IQ/OQ documentation the supplier could not provide, USD 15,000 on a serialization middleware integration that the machine’s non-standard PLC required, and USD 22,000 on a compliance rework after the first WHO inspection — for a net overspend of USD 20,000 compared to the compliant option, with a 9-month project delay.
For current HIJ Machinery configuration pricing on the DPP-250 thermoforming pharma blister packaging machine or the DPP-260 automatic blister packing machine, contact Forester’s team directly using the form below — pricing is project-specific and includes compliance documentation scope.
Frequently Asked Questions: Pharma Blister Packaging Market & Equipment
What is the current size of the global pharmaceutical blister packaging market?
The global pharmaceutical blister packaging market is valued at approximately USD 6.8 billion in 2024 and is projected to reach USD 9.2 billion by 2030, growing at a compound annual growth rate (CAGR) of 5.1%–6.3%. Asia-Pacific is the fastest-growing region at 7.2% CAGR, driven by generic drug volume expansion in India, Indonesia, Vietnam, and the Philippines. North America and Europe show slower growth (3.6%–4.3% CAGR) due to market maturity but remain the highest-value installed base for compliance-grade equipment upgrades triggered by EU FMD and DSCSA serialization mandates.
Why is the Alu-Alu cold-form blister segment growing faster than thermoforming?
Cold-form Alu-Alu blister packaging is growing at 8.4% CAGR versus 4.8% for thermoforming because moisture-sensitive drug formulations — including oncology generics, ACE inhibitors, and ARBs — require a water vapor transmission rate (WVTR) of less than 0.005 g/m²/day that only aluminum cold-form laminates can achieve. PVC/PVDC films at 90g coating weight deliver WVTR of approximately 0.2–0.5 g/m²/day — insufficient for Zone IVb ICH Q1A stability compliance in high-humidity markets such as Southeast Asia, coastal Brazil, and Gulf region countries. As generic drug production in these high-humidity markets accelerates, Alu-Alu adoption accelerates proportionally.
What compliance documentation should a pharmaceutical blister machine supplier provide?
A compliant pharmaceutical blister machine supplier must provide, at minimum: (1) CE marking certificate referencing ISO 13849-1 for machinery safety; (2) a cGMP compliance declaration referencing EU GMP Annex 1 (2022 revision) structural requirements; (3) Installation Qualification (IQ) and Operational Qualification (OQ) protocols per EU GMP Annex 15 §10.3, including sealing temperature uniformity test records (±2°C across the die face); (4) 21 CFR Part 11 §11.10(e)-compliant audit trail documentation confirming that all sealing parameter changes are logged with operator ID and timestamp; and (5) a FAT report signed by both supplier and customer. Suppliers who cannot provide items 3–5 as contractual deliverables — not “available on request” — are incompatible with WHO GMP, EU GMP, or FDA-regulated production environments.
How does DSCSA serialization affect blister packaging machine selection in 2025?
The US Drug Supply Chain Security Act (DSCSA), fully enforced from November 2024, requires that every saleable unit leaving a blister packaging line carries a unique 2D DataMatrix code encoding GTIN, lot number, expiry date, and serial number (SGTIN-96 format). This requirement demands that your blister machine’s PLC communicate with a serialization vision system with a trigger latency below 0.3 seconds per blister at line speed — achieved reliably only through PROFINET IO (4ms cycle time) or EtherNet/IP protocols, not legacy Modbus TCP (500ms polling). Machines without native high-speed PLC communication protocols require custom middleware integration costing USD 15,000–40,000 and adding 8–14 weeks to project timelines. Specify DSCSA serialization readiness as a mandatory URS requirement before any vendor engagement.
What is the typical ROI timeline for a pharmaceutical blister packaging machine investment?
Based on HIJ Machinery’s project analysis across 40+ installations, a mid-speed servo blister line (USD 85,000–160,000) in a regulated generic pharma facility typically achieves payback in 18–30 months when running at 85% OEE, producing batches of 500,000–1,000,000 tablets/shift. The ROI calculation is significantly affected by compliance architecture: a machine that fails FAT and requires a 3-month rework delays revenue generation and adds USD 30,000–80,000 in unplanned costs, extending payback to 36–48 months. Conversely, a turnkey line with bundled IQ/OQ/PQ documentation typically starts validated production 4–6 months faster than a point-solution purchase requiring third-party validation — recovering the USD 20,000–40,000 compliance bundle cost within the first 8 months of production.
Which markets are driving the fastest growth in pharmaceutical blister packaging equipment demand?
Southeast Asia (Indonesia, Vietnam, Philippines, Thailand), Latin America (Brazil, Colombia, Mexico), and the Middle East (Saudi Arabia, UAE, Egypt) are collectively the fastest-growing equipment demand markets for pharmaceutical blister packaging in 2024–2030, driven by three converging factors: (1) domestic generic drug production scale-up to reduce API import dependency; (2) WHO prequalification campaigns that require GMP-compliant packaging infrastructure; and (3) regional regulatory authorities (BPOM in Indonesia, ANVISA in Brazil, SFDA in Saudi Arabia) aligning their inspection criteria with EU GMP and WHO TRS 992 Annex 3 standards, forcing equipment upgrades across existing installed bases. HIJ Machinery actively services all three of these regions with dedicated after-sales engineering teams.
🎯 Turn Market Intelligence Into Your Next Compliant Blister Line
The market is growing at 5–6% CAGR. Your competitors are upgrading. Compliance enforcement is tightening. The buyers who win are those who move from market data to a validated, serialization-ready blister line faster — without the compliance rework that drains 18 months of ROI.
“At HIJ Machinery, I don’t just sell you a machine; I deliver project certainty. Every blister line we ship comes with cGMP documentation, FAT protocols, and a serialization architecture that survives your first regulatory audit — not just your first shift.” — Forester Xiang, Founder, HIJ Machinery
✅ IQ/OQ/PQ Documentation Included • ✅ Serialization-Ready (EU FMD & DSCSA) • ✅ 72-Hour Spare Parts Dispatch • ✅ On-Site FAT Support Available
🔗 Continue your research: visit the HIJ Machinery Knowledge Hub for in-depth technical guides on blister packaging compliance, machine selection, and validation protocols — or learn more about why global pharma buyers choose HIJ Machinery for mission-critical production lines.


